
SLC35B1 transports ATP into the ER, and its structure offers potential for targeted therapies in diseases linked to ER stress.
A team of scientists has solved a long-standing question in cell biology by uncovering how the cell’s primary energy source, ATP, is transported into the endoplasmic reticulum (ER). Disruptions in this energy transport process may contribute to diseases such as type 2 diabetes, cancer, and neurodegenerative disorders. The study, published in Nature, confirms that the transporter protein SLC35B1 serves as the main gateway for ATP to enter the ER.
The research, led by David Drew, Professor of Biochemistry at Stockholm University and based at SciLifeLab, provides the first structural and mechanistic insight into how ATP reaches the ER using SLC35B1. The ER acts as the cell’s main “shipping port,” where it packages proteins and lipids, performs quality control, and supports their movement within the cell. These critical functions all depend on a steady supply of ATP.
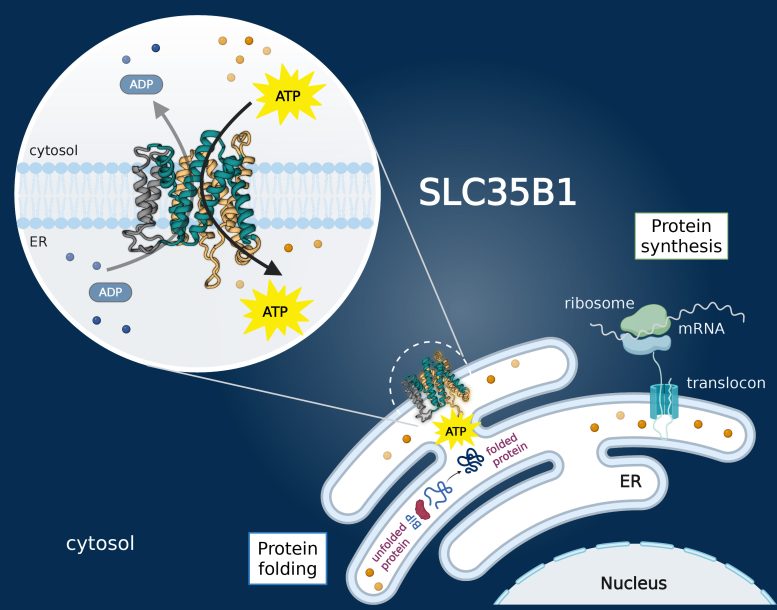
“Despite decades of research into ER function, the question of how ATP reaches the inside of the ER has been unclear. By confirming SLC35B1 as the transporter and resolving its structure with cryo-electron microscopy, we’ve not only answered a fundamental biological question, but also opened up new opportunities for therapeutic intervention”, says David Drew.
Future drug development
The findings have important implications for human health. Disrupted activity in the endoplasmic reticulum is associated with diseases such as type 2 diabetes, cancer, and neurodegenerative disorders, where ER stress and protein misfolding are key factors. Now that researchers have a detailed molecular blueprint of SLC35B1, the protein offers a promising target for the development of new drugs.

“Understanding how energy is delivered into the ER gives us powerful new ways to tackle a range of diseases that stem from ER dysfunction. Modulating SLC35B1 activity could become a new strategy for restoring ER balance in disease states”, says David Drew.
Large-scale screen
SLC35B1 has previously been proposed as an ATP transporter for the ER (Nature Comms 9:3489), but biochemical and structural validation was lacking. There have also been other, conflicting reports about its function. Teaming up with Giulio Superti-Furga Lab (CeMM, Austria), a large-scale CRISPR/Cas9 knockout screen of all SLC transporters showed that SLC35B1 was one of the five most essential transporters for cell growth, consistent with its proposed function.
Increasing protein size
An antibody against human SLC35B1 was then generated by Norimichi Nomura’s team (Kyoto Medical School, Japan), which was essential for increasing the size of the protein so it could be imaged by cryo-electron microscopy (cryo-EM). David Drew’s team then used the cryo-EM platform at SciLifeLab to visualize SLC35B1 in multiple conformations, revealing how it recognizes and transports ATP into the endoplasmic reticulum lumen. The structural data also highlighted critical amino acid residues involved in ATP binding and transport, suggesting potential sites for therapeutic targeting.
Targeted therapies
The team is currently screening for small molecules that modulate SLC35B1 function, with the goal of developing targeted therapies to either enhance or inhibit ATP transport when needed.
Reference: “Stepwise ATP translocation into the endoplasmic reticulum by human SLC35B1” by Ashutosh Gulati, Do-Hwan Ahn, Albert Suades, Yurie Hult, Gernot Wolf, So Iwata, Giulio Superti-Furga, Norimichi Nomura and David Drew, 21 May 2025, Nature.
DOI: 10.1038/s41586-025-09069-w
Funding: Knut och Alice Wallenbergs Stiftelse, Göran Gustafsson Foundation, AMED Basis for Supporting Innovative Drug Discovery and Life Science Research, H2020 The European Institute of Innovation and Technology, Japan Society for the Promotion of Science (JSPS)
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.