Researchers have identified two proteins on the surface of neurons that may help drive the spread of Parkinson’s disease in the brain.
New Yale School of Medicine (YSM) research suggests that two proteins on the surface of brain neurons involved in movement may play a key role in the progression of Parkinson’s disease.
Parkinson’s disease is a neurodegenerative disorder in which neurons gradually deteriorate and die. This cell loss is linked to the buildup of α-synuclein, a protein that becomes misfolded and can spread from one neuron to another.
Scientists still do not fully understand how α-synuclein moves between cells. A new study in Nature Communications points to two membrane proteins, mGluR4 and NPDC1, as important factors that help carry misfolded α-synuclein into healthy neurons after it is released by dying ones.

Senior author Stephen Strittmatter, MD, PhD, Vincent Coates Professor of Neurology and chair of the Department of Neuroscience at YSM, says the discovery could support the development of better Parkinson’s treatments.
Misfolded α-synuclein is “the pathologic hallmark of Parkinson’s disease,” he says. “If we understood how it gets into neurons, we could perhaps block or slow down the progression of the disease,” he adds. But to do that, “we need to understand the molecular mechanism of how it spreads.”
Transporting α-synuclein
Neurodegenerative diseases, including Alzheimer’s and Parkinson’s, are becoming an increasing health concern in the United States. The Parkinson’s Foundation estimates that about 1.1 million people in the U.S. are currently living with Parkinson’s disease, with nearly 90,000 new diagnoses each year.
Parkinson’s disease often causes movement-related symptoms, including tremors, balance problems, and slower movement. These symptoms are tied to the accumulation of misfolded α-synuclein in motor-related brain cells. As the protein spreads from neuron to neuron, symptoms become worse.
One possible way α-synuclein enters new cells is by attaching to proteins on the cell surface. To test that possibility, Strittmatter and his colleagues generated 4,400 groups of cells, each designed to express different surface proteins, and examined whether any of them bound to misfolded α-synuclein.
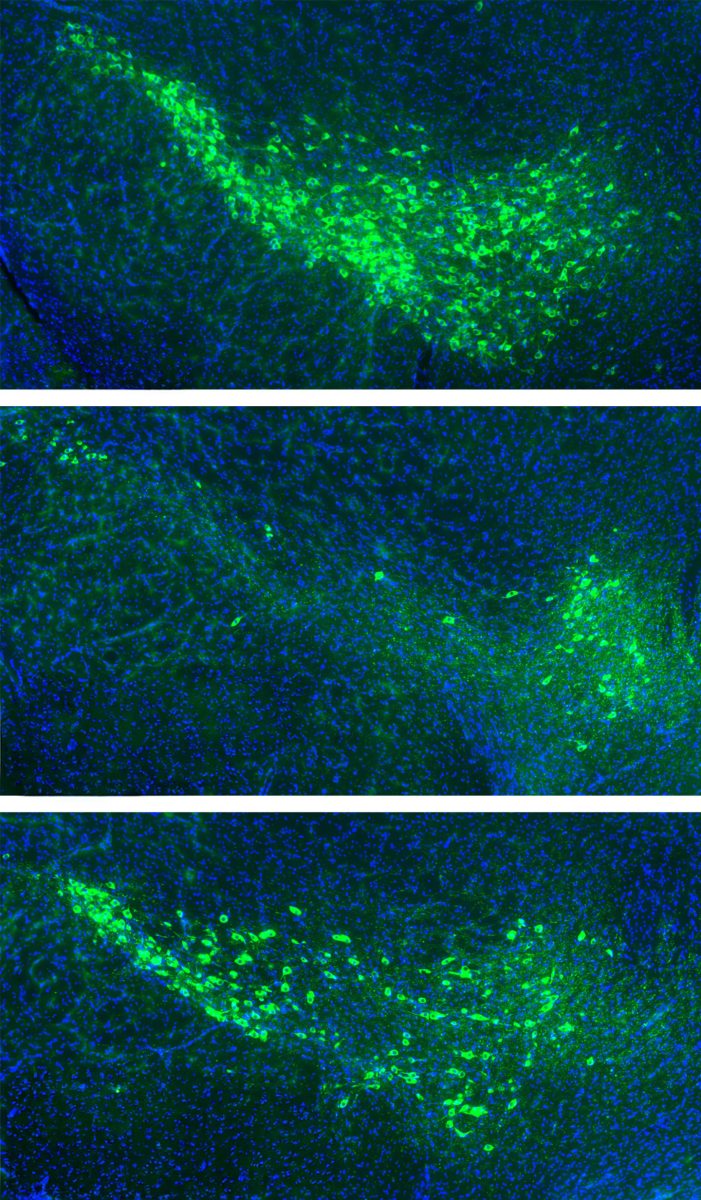
Most of the surface proteins did not bind to it. However, 16 did, including two found in human dopamine neurons in the substantia nigra, the brain region that degenerates in Parkinson’s disease. The researchers found that these two proteins, mGluR4 and NPDC1, carried misfolded α-synuclein into cells.
Preventing the spread of Parkinson’s pathology
The results led Strittmatter and his colleagues to suspect that mGluR4 and NPDC1 may help α-synuclein move between neurons. To investigate further, the researchers genetically engineered mice so that either mGluR4 or NPDC1 no longer functioned, then introduced misfolded α-synuclein.
In normal mice, the introduced misfolded α-synuclein built up in the brain, and the animals developed Parkinson ’s-like symptoms. Mice lacking functional mGluR4 or NPDC1 did not show the same pattern. The researchers also found that removing the genes for these two surface proteins in a mouse model of Parkinson’s disease reduced the risk of death and slowed symptom progression.
Together, the experiments suggest that mGluR4 and NPDC1 act together to help move misfolded α-synuclein into neurons in mice.
Strittmatter says the findings point to a possible new route for Parkinson’s disease treatment. Current therapies mainly help manage symptoms, but they do not effectively stop the disease from progressing. Targeting the spread of α-synuclein directly could lead to treatments that slow or possibly halt Parkinson’s disease, he says.
Such treatments could become increasingly important in the years ahead. Parkinson’s disease and other neurodegenerative conditions mainly affect older adults. As the number of Americans over age 65 rises in the coming decades, more people will face a higher risk of developing Parkinson’s disease.
“We have an aging population. How we can stop or slow neurons from dying is an enormous problem,” says Strittmatter. “This is really the time to make some inroads into figuring out how to slow it down.”
Reference: “mGluR4–NPDC1 complex mediates α-synuclein fibril-induced neurodegeneration” by Azucena Perez-Canamas, Mingming Chen, Leire Almandoz-Gil, Nabab Khan, Si Jie Tang, Allyson Ho, Erik C. Gunther and Stephen M. Strittmatter, 25 December 2025, Nature Communications.
DOI: 10.1038/s41467-025-67731-3
This work was supported by grant 16257 from the Michael J. Fox Foundation to A.P.C., and by grants R01AG034924, R35NS097283, R01AG070926, R01AG066165 and P30AG066508 from the N.I.H. to S.M.S.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
thanks for this