UCLA researchers have found that specific molecular changes in mice may actually provide protective effects instead of causing harm.
As people age, muscles take longer to recover after injury, a challenge that many older adults experience firsthand.
A study from UCLA using mice points to an unexpected explanation. Muscle stem cells in older tissue build up higher levels of a specific protein that slows their ability to activate and repair damage, yet at the same time helps them endure the more stressful conditions found in aging tissue.
Published in the journal Science, the findings indicate that some biological changes linked to aging may serve a protective role rather than being purely harmful. “This has led us to a new way of thinking about aging,” said Dr. Thomas Rando, senior author of the new study and director of the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research at UCLA.
“It’s counterintuitive, but the stem cells that make it through aging may actually be the least functional ones. They survive not because they’re the best at their job, but because they’re the best at surviving. That gives us a completely different lens for understanding why tissues decline with age.”
Protein buildup slows but protects cells
The team, led by postdoctoral scholars Jengmin Kang and Daniel Benjamin, analyzed muscle stem cells taken from both young and old mice. They found that levels of a protein called NDRG1 rose sharply with age, reaching about 3.5 times higher in older cells compared to younger ones. This protein acts as a brake inside the cell by suppressing the mTOR signaling pathway, which normally drives cell activation and growth.
To determine whether NDRG1 was behind the slower repair seen in older muscle, the researchers allowed mice to age naturally to roughly the equivalent of 75 human years, then inhibited the protein’s activity. Once NDRG1 was blocked, aged stem cells quickly regained youthful behavior, activating faster and improving muscle repair following injury.
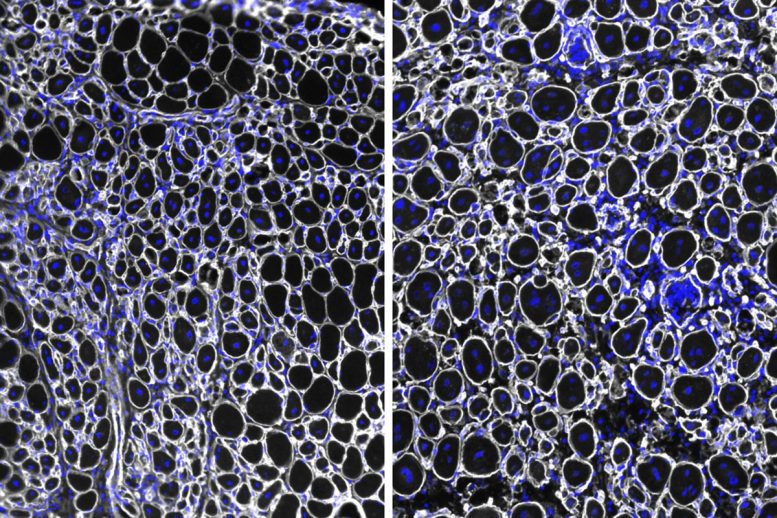
This improvement came with a drawback. Without the protective influence of NDRG1, fewer stem cells remained over time, reducing the tissue’s capacity to recover from repeated damage.
Trade-off between repair and survival
“Think of it like a marathon runner versus a sprinter,” said Rando, who is also a professor of neurology at the David Geffen School of Medicine at UCLA. “The stem cells in young animals are hyper-functioning — really good at what they do, namely sprinting, but they’re not good for the long term. They can make it through the 100-yard dash, but they can’t make it even halfway through the marathon. By contrast, aged stem cells are like marathon runners — slower to respond, but better equipped for the long haul. However, what makes them so proficient over long distances is exactly what renders them poor at sprinting.”
The researchers confirmed these results using several approaches, examining muscle stem cells from both young and aged mice in lab settings and within living tissue. Across all tests, higher NDRG1 levels were consistently linked to slower activation and repair, alongside greater cell survival and durability over time.
Survivorship bias shapes aging cells
The findings point to what the scientists describe as a “cellular survivorship bias.” Stem cells that fail to build up enough NDRG1 tend to die off as time passes, leaving behind a population that is more resistant but less efficient.
“Some age-related changes that look detrimental—like slower tissue repair—may actually be necessary compromises that prevent something worse: the complete depletion of the stem cell pool,” Rando said.
Rando compares this shift to survival strategies seen across the natural world. In extreme conditions such as droughts, famines, or freezing environments, organisms often prioritize survival over reproduction. In a similar way, stem cells appear to redirect resources away from producing new cells and toward maintaining their own survival as tissues age.
“Species survive because they reproduce, but in times of deprivation, animals turn on their own resilience programs,” Rando said. “There are a lot of examples in nature of allocating resources to survival under times of stress. It’s exactly aligned with what we’re seeing at the cellular level.”
Therapeutic gains come with trade-offs
These insights may inform future treatments aimed at improving tissue repair by adjusting the balance between stem cell activity and survival. Still, Rando emphasizes that any intervention is likely to involve trade-offs.
He states, “There’s no free lunch. We can improve the function of aged cells for a period of time, for certain tissues, but every time we do this, there’s going to be a potential cost and a potential downside.”
The research team plans to continue exploring the molecular mechanisms that govern this balance.
“This gene is almost like our doorway that we’ve opened into understanding what controls these trade-offs that are so critical, not only for evolution of species but also for the aging of tissues within an individual,” Rando said.
Reference: “Cellular survivorship bias as a mechanistic driver of muscle stem cell aging” by Jengmin Kang, Daniel I. Benjamin, Qiqi Guo, Chauncey Evangelista, Soochi Kim, Marina Arjona, Pieter Both, Mingyu Chung, Ananya K. Krishnan, Gurkamal Dhaliwal, Richard Lam and Thomas A. Rando, 29 January 2026, Science.
DOI: 10.1126/science.ads9175
The study was funded by the National Institutes of Health, the NOMIS Foundation, the Milky Way Research Foundation, the Hevolution Foundation and the National Research Foundation of Korea.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
14 Comments
Shimmer and shine zeta muscles growth
I want someone to find a cure for neuropathy
I had 2 rotary cuff 2 year a part each shoulder they will not heal in the inside cause chronic pain the that it have try every and can’t give a answer I 66 year old I don’t know else to do please help
I recommend painkillers lol Seriously, I just had my second rotator cuff reattachment in 15 years and I cannot stress enough to follow through with physical therapy, it really helps. If had the means ($) I would have had platelet therapy with the surgery. It’s criminal insurance companies won’t cover “cures”.
Hope you find comfort.
Another nothing-burger headline
Check into this it’s called EMSCULPT NEO it Builds Muscle and form & function. emsculptneo.com
They have a video out on people like you. My doctor suggested to my husband.
Have a blessed day and I pray this helps you and gives you relief.
Check into this it’s called EMSCULPT NEO it Builds Muscle and form & function. emsculptneo.com
They have a video out on people like you. My doctor suggested to my husband.
Have a blessed day and I pray this helps you and gives you relief.
Have you tried human growth hormone therapy?
I’m about too. There is too much good coming from it.
Go to YouTube
Type in Austin Gogh rotator cuff
Give thanks
Hi James, I am actively working on a rotary cuff injury suffered last December falling on a ice covered sidewalk. I am making slow but good progress. I am using a Tai Chi exetcise several times a day, then using a heat pad at night for the pain. Consult a phisotherapist for help. It will be a long process. I am 72.
I took my husband to get stem cell therapy in both shoulders and both torn biceps. Before we went, he could not lift anything without severe pain, and could not lift his arms to shoulder level). He was a very active man before his muscle issues/damage that happened over time from building and lifting sofa sleepers many years of his life. We went to Dr. Nathan Kellum, in Frankinton, TN –GREAT PHYSICIAN/SURGEON! After the stem cells (big procedure), my husband can now lift just about anything, raise his arms above his head, and lives his usual active life (well, not lifting sofa sleepers anymore). I highly recommend Dr. Kellum, and his stem cell clinic.
I hope you find relief!
Carmen
Carmen,
How long did it take your husband to see relief after the stem cell therapy on his shoulders and when did he have it done and is he still feeling good with it now.
Thank you
Lynn
If this is how cells react to aging, I’d love to know how they react to being chronically disabled at a young age as I’ve heard chronic pain can cause early onset dementia.