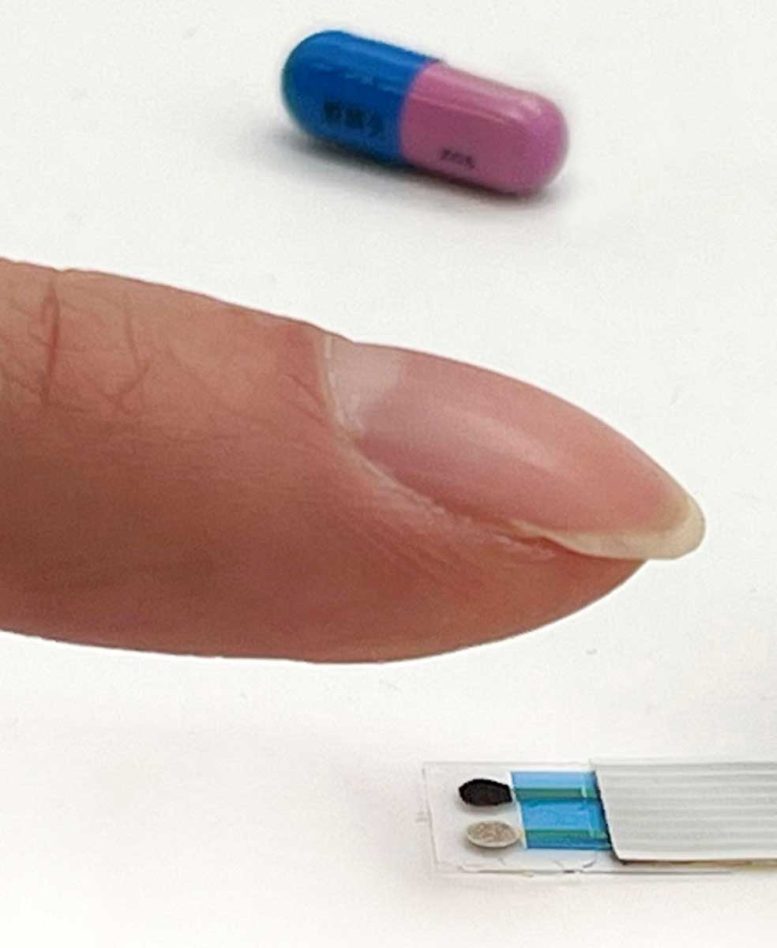
New Lithium-Sensing Technology for Bipolar Disorder Patients
If taken in just the right amount, lithium can alleviate the symptoms of bipolar disorder and depression. Too little won’t work, while too much can cause dangerous side effects. Patients must undergo invasive blood tests to precisely monitor the amount of this medication in the body. But today, researchers report the invention of a tiny sensor that detects lithium levels from sweat on the surface of a fingertip in as little as 30 seconds. It is very convenient and doesn’t require a trip to the clinic.
The scientists presented their results last week at the fall meeting of the American Chemical Society (ACS). ACS Fall 2022 featured nearly 11,000 presentations on a wide range of science topics.
Not only must lithium be taken at a specific dosage, but patients often struggle to take it as prescribed and may miss pills. This means that when the medication doesn’t appear to be working, health care providers need to know how much medication the patient is actually swallowing. However, current options for monitoring have significant drawbacks. For instance, blood draws produce accurate results, but they are invasive and time-consuming. Pill counters, meanwhile, don’t directly measure the intake of the medication. To address these limitations, the research team turned to another body fluid.
Sweat as a Non-Invasive Solution
“Although it may not be visible, the human body constantly produces sweat, often only in very small amounts,” says Shuyu Lin, Ph.D.. Lin is a postgraduate student researcher who co-presented the work with graduate student Jialun Zhu at the meeting. “Small molecules derived from medication, including lithium, show up in that sweat. We recognized this as an opportunity to develop a new type of sensor that would detect these molecules.”
“Through a single touch, our new device can obtain clinically useful molecular-level information about what is circulating in the body,” says Sam Emaminejad, Ph.D., the project’s principal investigator, who is at the University of California, Los Angeles (UCLA). “We already interact with a lot of touch-based electronics, such as smartphones and keyboards, so this sensor could integrate seamlessly into daily life.”
However, devising a sensor to detect lithium presented some technical challenges. Sweat is typically only present in minute amounts, but the electrochemical sensing needed to detect charged particles of lithium required an aqueous, or watery, environment. To provide it, the scientists engineered a water-based gel containing glycerol. This extra ingredient prevented the gel from drying out and created a controlled environment for the electronic portion of the sensor.
The team used an ion-selective electrode to trap the lithium ions after they traversed the gel. Ions accumulate generating a difference in electrical potential compared with a reference electrode. The scientists used this difference to infer the concentration of lithium present in sweat. Together, these components comprise a tiny, rectangular sensor that is smaller than the head of a thumbtack and can detect lithium in around 30 seconds. Although the sensor is still in the preliminary testing phase, ultimately, the research team envisions incorporating it into a larger, yet-to-be-designed system that provides visual feedback to the provider or the patient.
After characterizing the sensor using an artificial fingertip, the researchers recruited real people to test it, including one person on a lithium treatment regimen. The team recorded this person’s lithium levels before and after taking the medication. They discovered that these measurements fell close to those derived from saliva, which prior research has shown to accurately measure lithium levels. In the future, the scientists plan to study the effects of lotion and other skin products on the sensor’s readings.
Applications Beyond Lithium Monitoring
This technology also has applications beyond lithium. Emaminejad is developing similar touch-based sensors to monitor alcohol and acetaminophen, a painkiller also known by the brand-name Tylenol®, while also exploring the possibility of detecting other substances. The complete sensing systems could include additional features, such as encryption secured by a fingerprint, or, for substances that are often abused, a robotic dispensing system that releases medication only if the patient has a low level in their bloodstream.
The research team acknowledges support and funding from the National Science Foundation, Brain and Behavior Foundation, Precise Advanced Technologies and Health Systems for Underserved Populations, and the UCLA Henry Samueli School of Engineering and Applied Sciences.
Title
Touch-based non-invasive lithium monitoring using an organohydrogel-based sensing interface
Abstract
Lithium salt is one of the most widely-used psychiatric medications for individuals with bipolar disorder. Due to its narrow therapeutic window (~ 0.6 – 1.2 mM) and high nonadherence rate (~ 40%), it needs to be closely monitored to maximize the treatment efficacy. Standard practices of lithium monitoring for precise dosing are confined to centralized hospitals and involve invasive blood draw and high-cost lab-based analysis with long turnaround time. Moreover, currently there is no direct lithium adherence monitoring available, and the indirect monitoring solutions (e.g., pill counters) are incapable of verifying the actual intake event (inherently non-specific).
Overcoming these limitations, here, we developed a touch-based non-invasive lithium monitoring solution for decentralized lithium pharmacotherapy management. This solution is based on a hydrogel coated-sensing interface that collects and analyzes (in-situ) the flux of circulating lithium molecules that partition onto fingertips. This interface was constructed using a thin organohydrogel-coated lithium ion-selective electrode (TOH-ISE), where the TOH coating was specially engineered to render stabilized conditions for sensing. In particular, by adopting a water-glycerol bi-solvent matrix, the gel was endowed with anti-dehydration property (negligible weight loss for > 2 weeks storage in an ambient environment), resolving the dehydration challenge of previously-reported hydrogel-based interfaces. Furthermore, in the devised interface configuration, the TOH coating serves as a controlled micro-environment to condition the ISE in-situ; thus, it minimizes the ISE signal drift (a key challenge prohibiting the translation of ISEs in real-life applications).
To illustrate the clinical utility of our solution, the developed touch-based sensing interface was tested on a patient prescribed with lithium-based medicine, where the elevation of the circulating drug levels after the medicine intake was successfully captured. Collectively, our preliminary results demonstrate the suitability of our touch-based solution for lithium adherence monitoring, and more broadly for managing lithium-based pharmacotherapy.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.