
Using ‘patchy particles’ as a model for graphene’s atomic structure, researchers have gained novel insights into the formation and evolution of defects in two-dimensional materials. This groundbreaking study uncovers the early formation and subsequent repair of the most common defect, which has important implications for future engineering and application of these materials.
Graphene stands unrivaled in terms of strength among all known materials. In addition to its unparalleled robustness, its superior conductivity of heat and electricity makes it an incredibly versatile and unique material. The unprecedented properties of graphene were so remarkable that its discovery was honored with the Nobel Prize in Physics in 2010. However, our comprehension of this material and its related substances remains largely incomplete, primarily due to the immense challenge in observing the atoms that constitute them. To overcome this obstacle, a collaborative research effort from the University of Amsterdam and New York University has discovered an unexpected solution.
Materials that exist in two dimensions, composed of an ultra-thin, singular layer of atomic crystals, have been receiving considerable interest in recent times. This heightened interest is largely attributed to their atypical attributes, which significantly differ from their three-dimensional ‘bulk’ counterparts.
Graphene, the most famous representative, and many other two-dimensional materials, are nowadays researched intensely in the laboratory. Perhaps surprisingly, crucial to the special properties of these materials are defects, locations where the crystal structure is not perfect. There, the ordered arrangement of the layer of atoms is disturbed and the coordination of atoms changes locally.
Visualizing Atoms
Despite the fact that defects have been shown to be crucial for a material’s properties, and they are almost always either present or added on purpose, not much is known about how they form and how they evolve in time. The reason for this is simple: atoms are just too small and move too fast to directly follow them.
In an effort to make the defects in graphene-like materials observable, the team of researchers, from the UvA-Institute of Physics and New York University, found a way to build micrometer-size models of atomic graphene. To achieve this, they used so-called ‘patchy particles’.
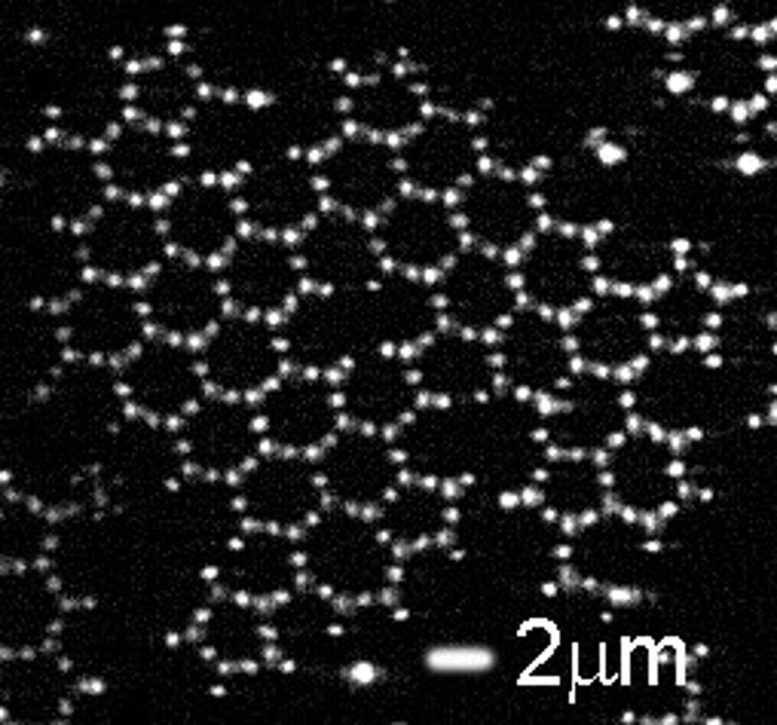
These particles – large enough to be easily visible in a microscope, yet small enough to reproduce many of the properties of actual atoms – interact with the same coordination as atoms in graphene, and form the same structure. The researchers built a model system and used it to obtain insight into defects, their formation, and evolution with time. Their results were recently published in the scientific journal Nature Communications.
Building Graphene
Graphene is made up of carbon atoms that each have three neighbors, arranged in the well-known ‘honeycomb’ structure. It is this special structure that lends graphene its unique mechanical and electronic properties. To achieve the same structure in their model, the researchers used tiny particles made of polystyrene, decorated with three even tinier patches of a material known as 3-(trimethoxysilyl)propyl – or TPM for short.
The configuration of the TPM patches mimicked the coordination of carbon atoms in the graphene lattice. The researchers then made the patches attractive so that the particles could form bonds with each other, again in analogy with the carbon atoms in graphene.
After being left alone for a few hours, when observed under a microscope the ‘mock carbon’ particles turned out to indeed arrange themselves into a honeycomb lattice. The researchers then looked in more detail at defects in the model graphene lattice. They observed that also in this respect the model worked: it showed characteristic defect motifs that are also known from atomic graphene. Contrary to real graphene, the direct observation and long formation time of the model now allowed the physicists to follow these defects from the very start of their formation, up to the integration into the lattice.
Unexpected Results
The new look at the growth of graphene-like materials immediately led to new knowledge about these two-dimensional structures. Unexpectedly, the researchers found that the most common type of defect already forms in the very initial stages of growth, when the lattice is not yet established. They also observed how the lattice mismatch is then ‘repaired’ by another defect, leading to a stable defect configuration, which either remains or only very slowly heals further to a more perfect lattice.
Thus, the model system not only allows to rebuild the graphene lattice on a larger scale for all sorts of applications, but the direct observations also allow insights into atomic dynamics in this class of materials. As defects are central to the properties of all atomically thin materials, these direct observations in model systems help further engineer the atomic counterparts, for example for applications in ultra-lightweight materials and optical and electronic devices.
Reference: “Visualizing defect dynamics by assembling the colloidal graphene lattice” by Piet J. M. Swinkels, Zhe Gong, Stefano Sacanna, Eva G. Noya and Peter Schall, 18 March 2023, Nature Communications.
DOI: 10.1038/s41467-023-37222-4
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
7 Comments
I believe your photo of the lattice should have shown the distance bar as nanometers, not micrometers!
Sorry! Ignore my previous comment.
I have been reading about the wonders of graphene for years now. Is it being used in any actual commercial products yet? And if so, how is it performing?
Very interesting for future humanity
Interesting photo. I always thought that Graphene was a collection of benzene rings consisting of six carbon atoms as depicted in the top photo. The second picture shows rings with what appears to be 12 particles. Interesting.
Upon further investigation, there appears to be a ring consisting of 10 particles, which form a pentagon. this would indicate that perhaps Graphene can arrange itself in a configuration similar to the patterns found on a soccer ball. I’ll have to look at Bucky balls to see how they are arranged. Very interesting.
Apparently Buckminsterfullerene, (Bucky ball), does in fact consist of atoms arranged in a pattern similar to a soccer ball. However, all of the depictions show a collection of 6 and 5 atom rings. The second photo shows collections of 12 and 10 particles, which I assume to be carbon atoms. Very, very, interesting.