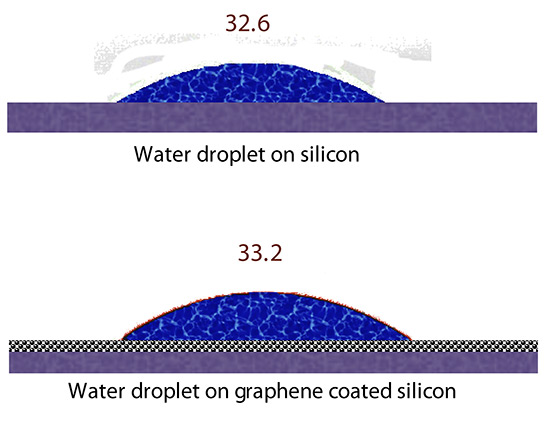
Graphene is the thinnest known material. In fact, according to a new study, it is so thin that it is essentially invisible to water when a single layer of graphene is used to cover silicon or most metals. The water behaves as if the graphene layer isn’t even there at all.
Rice University, Rensselaer study reveals graphene enhances many materials, but leaves them wettable. In short, graphene is largely transparent to the eye and, as it turns out, largely transparent to water.
A new study by scientists at Rice University and Rensselaer Polytechnic Institute (RPI) has determined that gold, copper and silicon get just as wet when clad by a single continuous layer of graphene as they would without.
The research, reported this week in the online edition of Nature Materials, is significant for scientists learning to fine-tune surface coatings for a variety of applications.
“The extreme thinness of graphene makes it a totally non-invasive coating,” said Pulickel Ajayan, Rice’s Benjamin M. and Mary Greenwood Anderson Professor in Mechanical Engineering and Materials Science and of chemistry. “A drop of water sitting on a surface ‘sees through’ the graphene layers and conforms to the wetting forces dictated by the surface beneath. It’s quite an interesting phenomenon unseen in any other coatings and once again proves that graphene is really unique in many different ways.” Ajayan is co-principal investigator of the study with Nikhil Koratkar, a professor of mechanical, aerospace and nuclear engineering at RPI.
A typical surface of graphite, the form of carbon most commonly known as pencil lead, should be hydrophobic, Ajayan said. But in the present study, the researchers found to their surprise that a single-atom-thick layer of the carbon lattice presents a negligible barrier between water and a hydrophilic – water-loving – surface. Piling on more layers reduces wetting; at about six layers, graphene essentially becomes graphite.
An interesting aspect of the study, Ajayan said, may be the ability to change such surface properties as conductivity while retaining wetting characteristics. Because pure graphene is highly conductive, the discovery could lead to a new class of conductive, yet impermeable, surface coatings, he said.
The caveat is that wetting transparency was observed only on surfaces (most metals and silicon) where interaction with water is dominated by weak van der Waals forces, and not for materials like glass, where wettability is dominated by strong chemical bonding, the team reported.
But such applications as condensation heat transfer — integral to heating, cooling, dehumidifying, water harvesting and many industrial processes — may benefit greatly from the discovery, according to the paper. Copper is commonly used for its high thermal conductivity, but it corrodes easily. The team coated a copper sample with a single layer of graphene and found the subnanometer barrier protected the copper from oxidation with no impact on its interaction with water; in fact, it enhanced the copper’s thermal effectiveness by 30 to 40 percent.
“The finding is interesting from a fundamental point of view as well as for practical uses,” Ajayan said. “Graphene could be one of a kind as a coating, allowing the intrinsic physical nature of surfaces, such as wetting and optical properties, to be retained while altering other specific functionalities like conductivity.”
Reference: “Wetting transparency of graphene” by Javad Rafiee, Xi Mi, Hemtej Gullapalli, Abhay V. Thomas, Fazel Yavari, Yunfeng Shi, Pulickel M. Ajayan and Nikhil A. Koratkar, 22 January 2012, Nature Materials.
DOI: 10.1038/nmat3228
The paper’s co-authors are Rice graduate student Hemtej Gullapalli, RPI graduate students Javad Rafiee, Xi Mi, Abhay Thomas and Fazel Yavari, and Yunfeng Shi, an assistant professor of materials science and engineering at RPI.
The Advanced Energy Consortium, National Science Foundation and the Office of Naval Research graphene MURI program funded the research.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
How does graphene respond to van der Waals forces? For example, how would a gecko walk up a graphene coated copper plate? Would it adhere to the surface as if the graphene were not there? Or would it fail to cling? Or would the graphene get all gummed up on the nanobristles of the gecko’s toe pads? Would a gecko with graphene coated toes still save me 15% on my car insurance?