Researchers targeted a common sodium ion channel to reverse pain, with positive results that could lead to a non-addictive solution to treat pain.
Researchers at the University of Arizona Health Sciences are closer to developing a safe and effective non-opioid pain reliever after a study showed that a new compound they created reduces the sensation of pain by regulating a biological channel linked to pain.
Most people experience pain at some point in their lives, and the National Institutes of Health estimates 100 million people in the U.S. suffer from chronic pain. Approximately 21-29% of patients prescribed opioids for chronic pain misuse them and 8-12% of people using an opioid for chronic pain develop an opioid use disorder, according to the National Institute on Drug Abuse. In 2019, nearly 50,000 people in the U.S. died from opioid-involved overdoses.
“Drug discovery for chronic pain is at the forefront of this research, and it’s being amplified by the intersection of the COVID-19 pandemic and the opioid epidemic,” said Rajesh Khanna, PhD, associate director of the UArizona Health Sciences Comprehensive Pain and Addiction Center and professor of pharmacology in the UArizona College of Medicine – Tucson. “Drug discovery is a very arduous process. Our lab looked at a fundamental mechanism of pain, came up with a way to differentiate it from those before us, and found a compound that has the potential as a new non-opioid treatment for pain.”

The paper, “Selective targeting of NaV1.7 via inhibition of the CRMP2-Ubc9 interaction reduces pain in rodents,” was published today (November 10, 2021) in Science Translational Medicine.
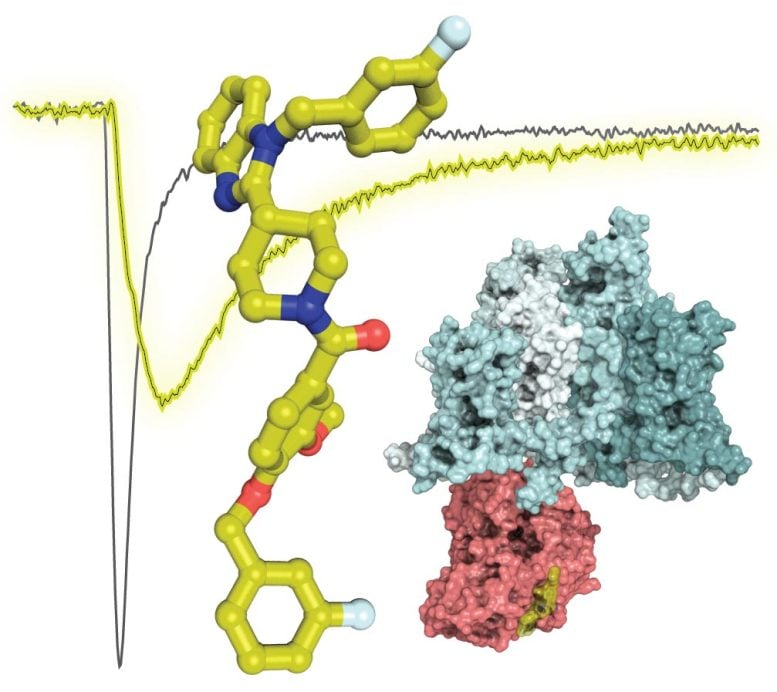
The biological mechanism at the heart of the research is NaV1.7, a sodium ion channel that previously was linked to the sensation of pain through genetic studies of people with rare pain disorders.
Nerve cells, or neurons, use electrical currents to send signals to the brain and throughout the body, and sodium ion channels are vital to a cell’s ability to generate those electrical currents. When a neuron is stimulated, the NaV1.7 channel opens and allows positively charged sodium ions to cross the cell membrane and enter the previously negatively charged cell. The change in charge across the cell membrane generates an electrical current, which increases the excitability of the neuron and sets in motion a cascade of events that leads to pain.
Because NaV1.7 is a human-validated target for pain, multiple attempts have been made to stop pain by using sodium ion channel inhibitors to block NaV1.7. None have been successful. Dr. Khanna and his team took a different approach – rather than block NaV1.7, they wanted to indirectly regulate it.
Using a compound they designed and dubbed 194, the team successfully regulated NaV1.7 activation in the laboratory using nerve cells from four different species, including humans. In animal models, 194 was effective in reversing pain in six different pain models in both sexes.
Researchers also found that 194 also may promote pain relief by activating the body’s endogenous, or naturally occurring, opioid system. Once produced, endogenous opioids activate receptors that produce physiological changes such as pain relief. And 194 did so without causing motor performance issues, depressive behaviors or addiction.
Finally, Dr. Khanna and the team observed a synergistic effect when 194 was combined with morphine and gabapentin. This is a promising sign that 194 could also be used in a dose-reduction strategy for painkillers that have negative side effects, including opioids, while maintaining high levels of pain relief.
The Science Behind 194
Dr. Khanna’s prior research identified a protein, collapsin response mediator protein 2 (CRMP2), and an enzyme, Ubc9, that both play a role in NaV1.7 activation. CRMP2 is a protein that binds to NaV1.7 and transports it to the cell membrane, where sodium ions are then transferred into the cell. Ubc9 is an enzyme that tags CRMP2 with another protein – a small ubiquitin-like modifier protein – to specifically direct control of NaV1.7.
Building on this knowledge, Dr. Khanna and the team set out to determine if they could directly regulate the activity of NaV1.7 by blocking Ubc9 from interacting with CRMP2. Team members including May Khanna, PhD, associate professor of pharmacology and BIO5 Institute member, Vijay Gokhale, PhD, associate research professor in the BIO5 Institute, and Samantha Perez-Miller, PhD, researcher and scientist in the Department of Pharmacology, examined 50,000 existing small molecules to identify the ones with a structure similar to Ubc9.
They selected less than 50 of the closest matches, which were then tested in Dr. Khanna’s laboratory to see if their presence would suppress the influx of sodium through NaV1.7. The findings were promising, so the team set their sights on developing a unique, more effective compound.
The result was 194, which UArizona patented and licensed to startup Regulonix LLC through Tech Launch Arizona, the UArizona office that commercializes inventions stemming from university research. Drs. Khanna and Gokhale founded Regulonix LLC in 2016 to address the growing opioid epidemic by developing new, non-addictive ways to treat pain and commercializing those innovations.
While 194 shows great promise for pain relief, Dr. Khanna and the team have been working with the National Institutes of Health’s National Center for Advancing Translational Sciences to optimize the compound. In this case, an NCATS team is primarily focusing on improving 194’s half-life – the time it takes for a drug to reduce by half in your body – and its drug-like properties.
It is an important step in optimizing the compound’s potential as a pain-relieving drug and advancing to the next stage, where researchers will file for Food and Drug Administration approval to begin clinical trials.
Reference: “Selective targeting of NaV1.7 via inhibition of the CRMP2-Ubc9 interaction reduces pain in rodents” by Song Cai, Aubin Moutal, Jie Yu, Lindsey A. Chew, Jörg Isensee, Reena Chawla, Kimberly Gomez, Shizhen Luo, Yuan Zhou, Aude Chefdeville, Cynthia Madura, Samantha Perez-Miller, Shreya Sai Bellampalli, Angie Dorame, David D. Scott, Liberty François-Moutal, Zhiming Shan, Taylor Woodward, Vijay Gokhale, Andrea G. Hohmann, Todd W. Vanderah, Marcel Patek, May Khanna, Tim Hucho and Rajesh Khanna, 10 November 2021, Science Translational Medicine.
DOI: 10.1126/scitranslmed.abh1314
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
9 Comments
I might feel like this could have merit were it not for the fact that the %’s of patients who become addicted are grossly over stated. Any time a potential treatment can’t be talked about without pushing inaccurate data I am going to automatically be suspicious of it. If they dont use accurate stats for one thing whos to say they are telling the truth about their new treatment
How could you accurately show the number of people whom are addicted to opiates? Some people don’t even say they’re addicted and they really are. It’s impossible to get 100% say on what the actual number is. I guess you’re probably suspicious of most medicines and studies out there.
Glad research is progressing, but soooo disheartened by the gross overstatement of statistics re: Opioid Use Disorder and legitimate pain patient misuse of their meds. Perpetuating the lies about the causes of the alleged “opioid crisis” does great damage. Why not just be honest and focus on the potential good of this breakthrough?!? Stats and comments grossly conflict with other, TRUE data.
How could you accurately show the number of people whom are addicted to opiates? Some people don’t even say they’re addicted and they really are. It’s impossible to get 100% say on what the actual number is. I guess you’re probably suspicious of most medicines and studies out there.
Who are you to judge that?!? Many people, doctors included, are equating physical dependence with addiction and that is INCORRECT.
New research is always wonderful news however the constant equating of chronic pain patients with the “opioid epidemic” is disheartening and misleading.
As a follow-up to my comment above, the article interested in learning about the UArizona Health Sciences Comprehensive Pain and Addiction Center. Their web page description, below, is regretfully just another example of equating chronic pain with addiction. Perhaps they should separate what are two very different conditions. Truly hope their research holds promise for those with chronic pain diseases and injuries.
————-
Comprehensive Pain and Addiction Center
OVERVIEW
Comprehensive Pain and Addiction CenterTo lead the state in addressing the current opioid epidemic, we will create a Comprehensive Center for Chronic Pain and Addiction that is the nucleus for treatment, research, trials, drug discovery, technology, education and legislation to predict, prevent and contain addiction crises. The university will conduct new research on comprehensive behavioral therapies as addiction treatment, develop early screeners for individuals at risk and develop novel non-addictive pain medications and medications to help those who suffer from addiction.
This is absolutely pointless. The entire issue with the pharmaceutical industry is there idea that because opioids are addictive and make you feel good. That that is inherently bad. They think they need to completely wipe the abuse potential away from everything and their spending billions on making drugs like this that I guarantee aren’t even tested on humans yet and even though they want to demonize opiates. They are a central need for any surgery without the body going into shock due to pain. We already have effective pain killers, we don’t need new ones. The last time they came out with what they called a non addictive painkillerto replace morphine. OxyContin happened and realized it actually was stronger and more addictive. The time they did that before that was when they were using Heroin over the counter as a cough medicine and flu medicine in the 1920s, then they said they had a new non addictive painkillerto once they realized how addictive Heroin was, and what was their pushed non addictive option? Morphine. See where I’m going. It’s useless we already have good painkillers get over the fact that people abuse them that’s nobodys concern if someone takes opiates illegally to self medicate.. I don’t think they should be demonized for it
The article’s stats are so far off, it is laughable. And the way the writer equates chronic pain with addiction is just showing what a sub-par journalist they really are. Pain patients don’t feel a euphoria with their legally prescribed medications – “the drug goes to the pain, not to the brain,” in case you never heard that saying. It’s very insulting that they conflate apples and oranges here, making the pain patients seem like addicts for needing the meds to have even a minimally decent quality of life.