
The cell model of ion-exchange membranes reveals violations of the Onsager reciprocity principle, particularly at high electrolyte concentrations, highlighting the importance of accounting for asymmetric transport coefficients.
Ion-exchange membranes are widely used in electrochemical and separation processes, and accurate modeling of their transport properties is essential. A new study published in the journal Asymmetry demonstrates that the commonly used cell model for ion-exchange membranes violates the Onsager reciprocity principle, challenging a foundational assumption in non-equilibrium thermodynamics.
In the context of the cell model for ion-exchange membranes, it is shown that the Onsager reciprocity principle does not hold, the cross kinetic coefficients are not equal. This violation arises because the reciprocity principle is valid only for systems in which all generalized fluxes vanish when thermodynamic forces other than zero are present, as stipulated by linear irreversible thermodynamics.
The cell model simplifies the complex structure of real ion-exchange membranes by replacing randomly distributed ion-exchange resin grains with a periodic lattice. This lattice consists of identical, porous, charged spheres surrounded by concentric spherical shells filled with electrolyte, forming a structured porous medium.
To account for interactions between neighboring particles, the model imposes specific boundary conditions at the outer surface of the liquid shell. It is assumed that the gradients of external forces acting on the entire porous layer are equivalent to the local gradients within a single unit cell.
Advantages of the Cell Model Approach
The advantage of the described approach is that all the quantities included in the equations of electrolyte transfer through a porous layer: thermodynamic fluxes and forces, can be directly measured in experiments.
When calculating the kinetic coefficients Lij of the Onsager matrix, as independent thermodynamic forces that are set during the experiment of transferring an electrolyte solution through a thin infinitely extended charged porous layer (membrane), we select gradients of pressure dp/dx, chemical dμ/dx and electrical dφ/dx potentials perpendicular to the surface of this layer.
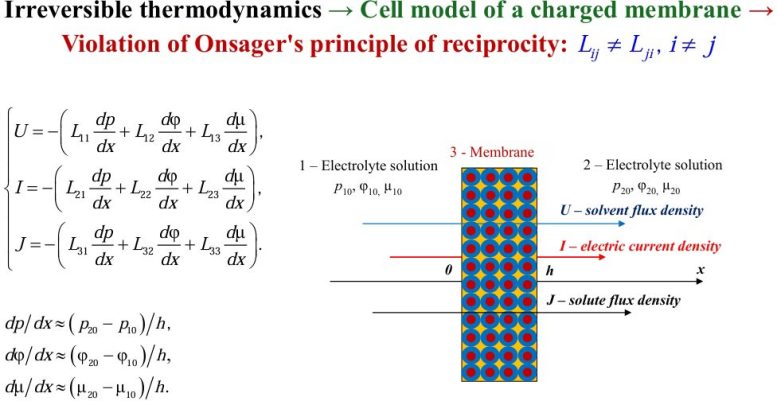
As dependent thermodynamic parameters determined in the experiment, we take the flux densities: U—solvent (for example, water), I—mobile charges (electric current density), J—solute (density of the diffusion flux of the electrolyte).
Experimental Observations
It was shown based on experiments for heterogeneous MK-40 and homogeneous MF-4SK cation-exchange membranes that their cross coefficients differ little at small concentrations of aqueous solutions of NaCl electrolyte (up to 0.1 M).
At high concentrations, there is a significant quantitative and qualitative discrepancy between reverse osmotic L13 and capillary osmotic L31 coefficients, as well as for electrodiffusion coefficients L23 and L32. At the same time, the cross kinetic coefficients L12 (regulates electroosmosis) and L21 (regulates streaming current) practically coincide up to high values of the electrolyte concentration.
We have shown here that in the case of the cell membrane model, the Onsager reciprocity principle is violated—the matrix of kinetic coefficients is not symmetric. In this regard, it is necessary to be careful about determining the transport characteristics of membranes, which depend on the cross kinetic coefficients due to the asymmetry of the latter.
In the present paper, it is shown that the coupled cross coefficients can differ not only quantitatively, but also qualitatively. It is crucial when considering the transport characteristics of charged membranes.
Reference: “Asymmetry of cross kinetic coefficients in the cell model of a charged membrane” by Anatoly Filippov, 11 April 2025, Asymmetry.
DOI: 10.55092/asymmetry20250003
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
14 Comments
Thermodynamics is always right. Try fixing your model instead.
This isn’t about villain of energy conservation, but the Onsager profile of reciprocity, which stress not seen to hold up at levels such as ion transfer.
Ugh…. Looks like it’s a fake paper. A lot of these scientific papers gets released with zero working models.
Anyone remember cold fusion and billions that got dumped down the toilet over bad released research papers promising results but never delivered…
The paper has models – did you read it?
Science and commercialization has produced much more value than bad papers and optimistic (and not always earnest) attempts to earn money on hard working scientist results have wasted.
🤣 Lord of the Rings verbage with Star Treck principles! I love it!
It’s a month after April fools’. Declaring thermodynamic violations isn’t most click bait, it’s jumping the shark entirely.
Note this due seem to have application, but the title is very deliberately misleading.
Not really, if you check e.g. Wikipedia or Google’s AI search overview, the former describe when it is supposed to be violated and the latter too in addition to a list of putative common situations of violation.
“Examples of Onsager Reciprocity Principle Violations:
Ion-exchange membranes:
In a cell model of ion-exchange membranes, the Onsager reciprocity principle is violated, meaning the cross-kinetic coefficients for reverse osmosis and capillary osmosis are not equal, especially at high concentrations.
Condensed matter physics:
Violations of the principle have been observed in underdoped cuprates, where measurements of the Nernst coefficient, anisotropy of conductivity, and thermopower indicate a breakdown of the reciprocity relationship.
Anomalous Hall effect:
In some materials, the anomalous Hall effect can violate the Onsager reciprocal theorem, where the deflection of electrons is not constant regardless of current direction.”
(As I noted in my own response to the article, a macroscopic magnetic field often suffices to break microscopic reversibility.)
No they didn’t.
Yes, the author did show a theoretic violation. But that is supposed to happen if you go too far from when the reciprocity relations apply.
This is interesting but difficult to confirm that cross kinetic coefficient varies during ion transport through membrane.
More study required .
Someone broke the laws of thermodynamics? Send them to Kelvin prison and put them to heat death!
It is a purely theoretic analysis where the assumedly low perturbations of the symmetric equilibrium system is driven to high levels. For example, a magnetic field often suffice to break microscopic reversibility.
Nothing to see here, but the one author is correct in his attempt to check where the reciprocity (which proof is shown in e.g. Wikipedia) fails.
Good comments Torbjorn. It is good to read intelligent replies I get tired of reading the opinions of quibbling geeks.
Humans, wither scientist or religious tend to learn a concept and that concept sets up like concrete in their mind. Later potential conflicting or threatening views to this reality of theirs are violently rejected mentally. They are unable to hold two seemingly conflicting viewpoints in their mind at the same time. So one of them has to be immediately rejected to restore inner peace. This is how the expression of being hard headed comes from. It is also a source of bias, and not in the spirit of science or religion.
Keep an open mind, and be nice.