
A hidden transition between two forms of liquid water may explain its most puzzling properties.
Why is water most dense at 4°C (39.2°F)? Why is it so vital for the origin and survival of life? These basic questions might seem like they should have simple answers in textbooks, yet they have remained some of science’s most persistent mysteries.
After more than a decade of work, researchers now say they have uncovered the root cause of water’s unusual behavior. Their findings could change how water is explained in textbooks.
A research team led by Professor Kyung Hwan Kim from the Department of Chemistry at POSTECH (Pohang University of Science and Technology), working with Professor Anders Nilsson from the Department of Physics at Stockholm University, has directly observed water’s liquid-liquid critical point (LLCP). This phenomenon has been one of the most difficult challenges in science for decades. The study was published in Science.
Although water is one of the most studied substances, it is still not fully understood. One leading idea suggests that its strange properties come from the presence of a liquid-liquid critical point, a condition where two different liquid forms of water become indistinguishable.
The Challenge of “No-Man’s-Land”
Scientists have long predicted that if the LLCP exists, it would lie in a deeply supercooled range between about -40°C (-40°F) and -70°C (-94°F), often called “no-man’s-land.” To test this, researchers must study liquid water that stays unfrozen below -40°C (-40°F). However, in this range, water freezes so quickly that traditional measurement methods cannot capture it, making direct observation nearly impossible for decades.
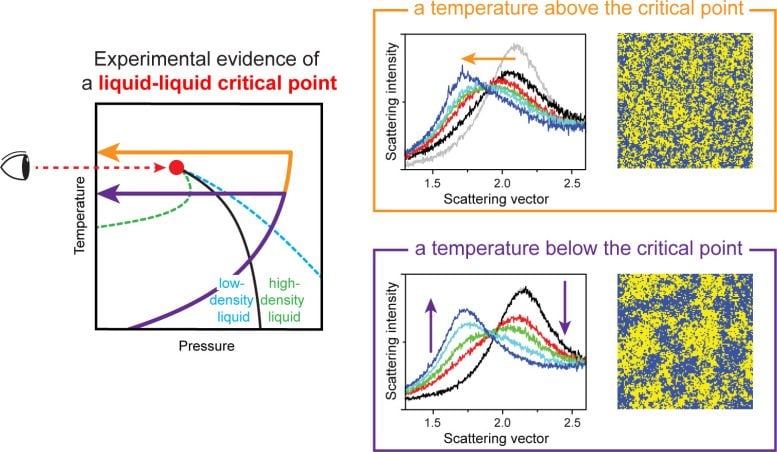
Over the past ten years, the team worked to overcome this barrier. They used an X-ray free-electron laser (XFEL), sometimes called “dream light,” which produces extremely intense X-ray pulses. This tool can capture molecular motion in as little as one ten-trillionth of a second. The experiments were carried out at PAL-XFEL at the Pohang Accelerator Laboratory.
Breakthroughs Over a Decade
In 2017, the researchers became the first to show that liquid water could be studied without freezing at temperatures as low as -45°C (-49°F). This proved that the once “inaccessible” region could be explored.
By 2020, they refined their method using amorphous ice, allowing them to extend measurements down to -70°C (-94°F). These experiments provided the first evidence that water can exist as two distinct liquid forms at extremely low temperatures. Both studies were published in Science and attracted wide attention.
Direct Observation of the Critical Point
In their latest work, the team closely tracked how water behaves under different temperatures and pressures. They report the first direct observation of a liquid-liquid critical point near -60°C (-76°F). At this point, two separate liquid states merge into a single supercritical liquid. This result reveals the underlying source of water’s unusual properties.
This discovery reflects years of steady effort focused on a fundamental scientific problem. By turning a long-standing theory into direct experimental evidence, the researchers have advanced our understanding of water.
Professor Kyung Hwan Kim said, “The intense debate in the scientific community, spanning many years, over water’s unusual properties and a liquid-liquid critical point has finally been brought to a close.” He added, “This discovery will serve as a starting point for uncovering the essential roles water plays in living systems and in a wide range of natural phenomena.”
Reference: “Experimental evidence of a liquid-liquid critical point in supercooled water” by Seonju You, Marjorie Ladd-Parada, Kyeongmin Nam, Aigerim Karina, Seoyoung Lee, Myeongsik Shin, Cheolhee Yang, Yeseul Han, Sangmin Jeong, Kichan Park, Kyeongwon Kim, Minjeong Ki, Robin Tyburski, Iason Andronis, Keely Ralf, Jae Hyuk Lee, Intae Eom, Minseok Kim, Rory Ma, Dogeun Jang, Fivos Perakis, Peter H. Poole, Katrin Amann-Winkel, Kyung Hwan Kim and Anders Nilsson, 26 March 2026, Science.
DOI: 10.1126/science.aec0018
This research was supported by the National Research Foundation of Korea (NRF), through the Outstanding Young Scientist Grant program and the Leading Research Center Support Program, and by the Samsung Science and Technology Foundation.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
I read this entire article and didn’t see exactly any breakthroughs in why water is most dense at 39 degrees, which was the question I thought the article addressed. So I assume the “breakthrough” regards behavior of two states of liquid water at high pressure/low temperatures? And hydrogen bonding is STILL the reason water is most dense at that temperature?
I also didn’t understand how a critical point at much below zero relates to the critical density point at 4C above zero. Wasn’t explored at all in this article.