The zap-and-freeze approach let scientists observe ultrafast synaptic recycling in both mouse and human brain tissue, highlighting conserved molecular mechanisms.
The technique could help reveal why communication breaks down in Parkinson’s and guide new treatment strategies.
Breakthrough “Zap-and-Freeze” Imaging in Live Brain Tissue
Researchers at Johns Hopkins Medicine report that they have used a “zap-and-freeze” method to observe brain cell communication in real, living tissue taken from both mice and humans. The approach allowed them to capture interactions that are usually extremely difficult to see.
According to the team, the results of these experiments, which were funded by the National Institutes of Health and published today (November 24) in Neuron, may offer new clues about the biological triggers behind nonheritable forms of Parkinson’s disease.
The Parkinson’s Foundation notes that sporadic cases of the condition make up the majority of diagnoses. These cases involve problems at the tiny junction where one brain cell sends signals to the next. This signaling site, called a synapse, has long been a challenge for scientists to examine in detail, says Shigeki Watanabe, Ph.D., an associate professor of cell biology at Johns Hopkins Medicine and the study’s lead investigator.
“We hope this new technique of visualizing synaptic membrane dynamics in live brain tissue samples can help us understand similarities and differences in nonheritable and heritable forms of the condition,” Watanabe says. He adds that insights gained through this method may eventually contribute to the development of new treatment strategies for the disorder.
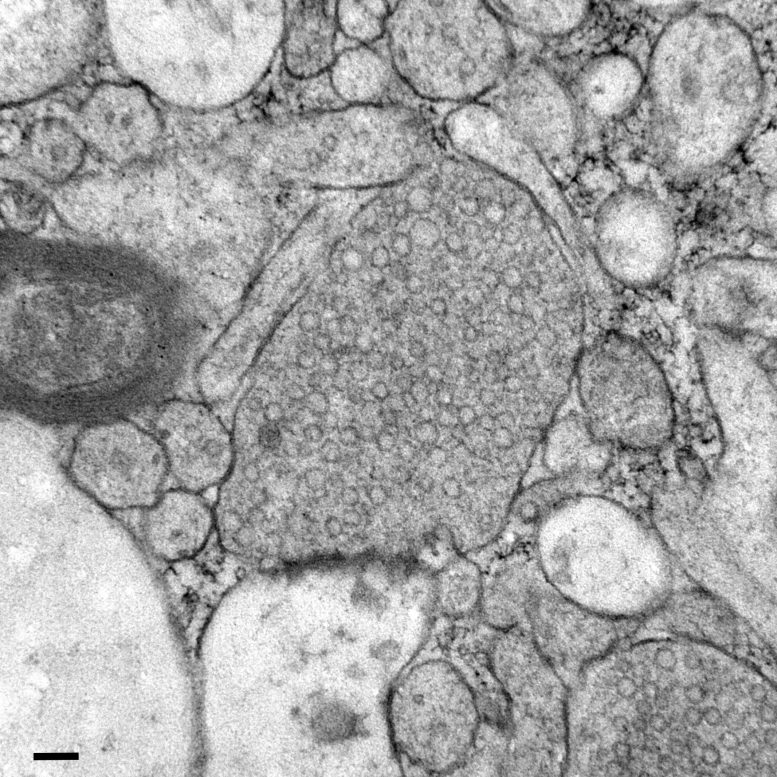
How Healthy Synapses Move Messages and Form Memories
In a healthy brain, synaptic vesicles act as tiny information carriers that help one cell pass messages to another. This activity is essential for tasks such as processing information, learning, and building memories. Watanabe says that understanding how these vesicles work is a key step in pinpointing where communication begins to falter in neurodegenerative diseases.
Watanabe previously helped create the zap-and-freeze technique to examine rapid changes in synaptic membranes (these results were published in 2020 in Nature Neuroscience). The method uses a brief electrical stimulus to activate living brain tissue, followed immediately by a fast freeze to preserve the movement of cellular structures for later viewing with electron microscopy.
In research published earlier this year in Nature Neuroscience, Watanabe applied the technique to genetically engineered mice to explore the role of a protein called intersectin. The study showed how intersectin maintains synaptic vesicles in a specific spot inside the cell until they are ready to be released and activate a nearby neuron.
Mouse and Human Brain Samples Provide Critical Insights
For the new study, the researchers used samples from the brains of normal mice as well as living cortical brain tissue sampled with permission from six individuals undergoing surgical treatment for epilepsy at The Johns Hopkins Hospital. The surgical procedures were medically necessary to remove lesions from the brain’s hippocampus.
Working with scientists Jens Eilers and Kristina Lippmann at Leipzig University in Germany, the researchers first validated the zap-and-freeze approach by observing calcium signaling, a process that triggers neurons to release neurotransmitters in living mouse brain tissues.
Watching Vesicle Fusion and Recycling in Real Time
Next, the scientists stimulated neurons in mouse brain tissue with the zap-and-freeze approach and observed where synaptic vesicles fuse with brain cell membranes and then release chemicals called neurotransmitters that reach other brain cells. The scientists then observed how mouse brain cells recycle synaptic vesicles after they are used for neuronal communication, a process known as endocytosis that allows material to be taken up by neurons.
The researchers then applied the zap-and-freeze technique to brain tissue samples from people with epilepsy, and observed the same synaptic vesicle recycling pathway operating in human neurons.
In both mouse and human brain samples, the protein Dynamin1xA, which is essential for ultrafast synaptic membrane recycling, was present where endocytosis is thought to occur on the membrane of the synapse.
“Our findings indicate that the molecular mechanism of ultrafast endocytosis is conserved between mice and human brain tissues,” Watanabe says, suggesting that the investigations in these models are valuable for understanding human biology.
Future Plans: Mapping Vesicle Dynamics in Parkinson’s Patients
In future experiments, Watanabe says he hopes to leverage the zap-and-freeze technique to study synaptic vesicle dynamics in brain tissue samples taken with permission from patients with Parkinson’s disease undergoing deep brain tissue stimulation.
Reference: “Ultrastructural membrane dynamics of mouse and human cortical synapses” by Chelsy R. Eddings, Minghua Fan, Yuuta Imoto, Kie Itoh, Xiomara McDonald, Jens Eilers, William S. Anderson, Paul F. Worley, Kristina Lippmann, David W. Nauen and Shigeki Watanabe, 24 November 2025, Neuron.
DOI: 10.1016/j.neuron.2025.10.030
Funding support for this research was provided by the National Institutes of Health (U19 AG072643, 1DP2 NS111133-01, 1R01 NS105810-01A1, R35 NS132153, S10RR026445), Howard Hughes Medical Institute, Kazato Foundation, American Lebanese Syrian Associated Charities, Marine Biological Laboratory, Leipzig University, Roland Ernst Stiftung, Johns Hopkins Medicine, Chan Zuckerberg Initiative, Brain Research Foundation, Helis Foundation, Robert J Kleberg Jr and Helen C Kleberg Foundation, McKnight Foundation, Esther A. & Joseph Klingenstein Fund, and the Vallee Foundation.
In addition to Watanabe, other scientists who contributed to this work include Chelsy Eddings, Minghua Fan, Yuuta Imoto, Kie Itoh, Xiomara McDonald, William Anderson, Paul Worley and David Nauen from Johns Hopkins, and Jens Eilers and Kristina Lippmann from Leipzig University, Germany.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.