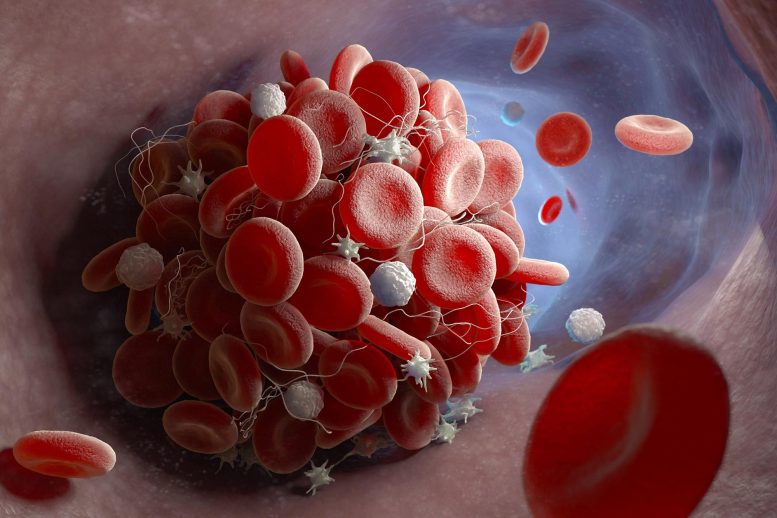
After studying blood samples from 244 patients hospitalized for COVID-19, a group of researchers, including those who work at the National Institutes of Health, identified “rogue antibodies” that correlate with severe illness and may help explain mechanisms associated with severe blood clotting. The researchers found circulating antiphospholipid antibodies, which can be more common among people with autoimmune disorders, such as lupus. However, these “autoantibodies,” which target a person’s own organs and systems, can also be activated in response to viral infections and activate other immune responses.
Scientists compared the blood samples to those from healthy controls and found the COVID-19 samples contained higher levels of the antibody IgG, which works with other immune cells, such as IgM, to respond to immune threats. Higher levels of IgG were also associated with COVID-19 disease severity, such as in patients who required breathing assistance. The researchers observed similar patterns, but to a lesser extent, after analyzing blood samples from 100 patients hospitalized for sepsis, which can leave the body in inflammatory shock following a bacterial or viral infection.
IgG helps bridge a gap between innate and adaptive immune responses – a process that helps the body recognize, respond to, and remember danger. In normal cases, these features help protect the body from illness and infection. However, in some cases, this response can become hyperextended or altered and exacerbate illness. A unique finding from this study is that when researchers removed IgG from the COVID-19 blood samples, they saw molecular indicators of “blood vessel stickiness” fall. When they added these same IgG antibodies to the control samples, they saw a blood vessel inflammatory response that can lead to clotting.
Since every organ has blood vessels in it, circulating factors that lead to the “stickiness” of healthy blood vessels during COVID-19 may help explain why the virus can affect many organs, including the heart, lungs, and brain. A query of this study was evaluating “upstream” factors involved with severe blood clotting and inflammation among people with severe COVID-19 illness.
The researchers note future studies could explore the potential benefits of screening patients with COVID-19 or other forms of critical illness for antiphospholipids and other autoantibodies and at earlier points of infection. This may help identify patients at risk for extreme blood clotting, vascular inflammation, and respiratory failure. Corresponding studies could then assess the potential benefits of providing these patients with treatments to protect blood vessels or fine-tune the immune system.
Reference: “Endothelial cell-activating antibodies in COVID-19” by Hui Shi MD, PhD, Yu Zuo MD, Sherwin Navaz BS, Alyssa Harbaugh BS, Claire K. Hoy BS, Alex A. Gandhi BS, Gautam Sule PhD, Srilakshmi Yalavarthi MS, Kelsey Gockman BS, Jacqueline A. Madison MD, Jintao Wang PhD, Melanie Zuo MD, Yue Shi PhD, Michael D. Maile MD, Jason S. Knight MD, PhD and Yogendra Kanthi MD, 17 February 2022, Arthritis & Rheumatology.
DOI: 10.1002/art.42094
Yogen Kanthi, M.D. is a cardiologist, Clinical Lasker Research Scholar, and leads the Laboratory of Vascular Thrombosis and Inflammation at the National Heart, Lung, and Blood Institute. He is also an assistant professor of cardiovascular medicine at the University of Michigan, Ann Arbor.



Be the first to comment on "Scientists Pinpoint “Rogue Antibodies” Associated With Severe COVID-19 Blood Clotting"