Worms reduce inflammation only on high-fiber diets; low fiber disables their benefits and disrupts gut health.
Intestinal worms may help calm inflammation in the human body, but only when they receive enough dietary fiber. Without sufficient fiber, they shift into a hibernation-like state and lose their protective benefits. This finding comes from a new study by parasitologists at the Biology Centre of the Czech Academy of Sciences, published in Nature Communications.
For most of human evolution, intestinal parasites were a normal part of the digestive system. In industrialized nations, they have largely disappeared in recent decades, mainly due to improved hygiene and modern medical care.
During the same period, cases of autoimmune and inflammatory bowel diseases have increased. About 20 years ago, researchers began exploring whether certain worms could be used to treat these conditions, an approach known as helminth therapy.

“However, the results of helminth therapy have been inconsistent—sometimes the worms suppress inflammation, sometimes they do not. That’s why we focused on factors that may influence their effects in the gut,” says Kateřina Jirků from the Institute of Parasitology, Biology Centre CAS. The researchers identified diet, especially fiber intake, as a key factor.
Experimental Model and Dietary Conditions
The scientists studied how an intestinal parasite responds to different levels of dietary fiber in its host. They used the rat tapeworm Hymenolepis diminuta, a species widely used to study interactions among parasites, gut microbes, and the immune system. This tapeworm does not cause disease and has previously shown anti-inflammatory properties.
“We found that when the diet contains a high proportion of structural fiber, the tapeworm is not only in excellent condition but is also able to induce an anti-inflammatory response in the host. When fiber is lacking, the worm enters an energy-saving state resembling hibernation in mammals, and its anti-inflammatory effect disappears,” explains Jirků.
When fiber was low, the worms stayed much smaller, did not reach sexual maturity, and failed to produce eggs. Genetic analysis showed major shifts in gene activity related to growth, metabolism, and reproduction.
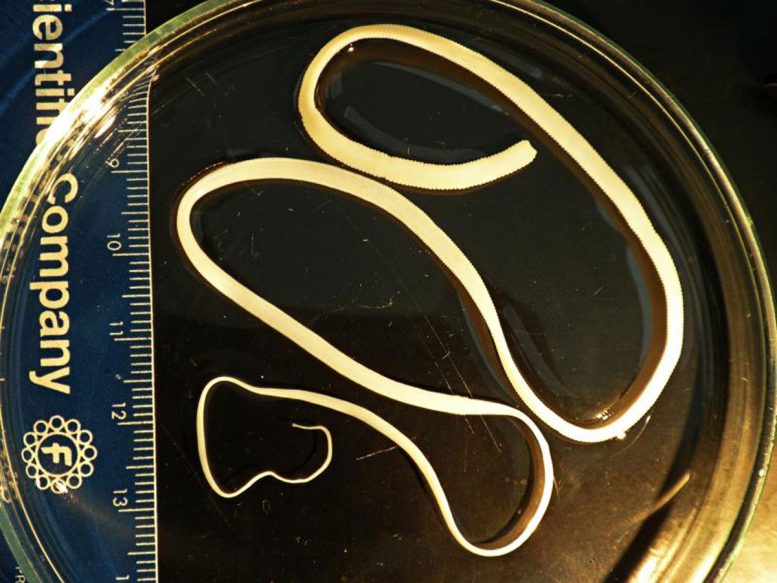
Gut Microbiome and Immune Response
Diet also strongly influenced the host’s gut microbiome. A fiber-rich diet supported beneficial bacteria linked to a healthy gut, while a Western-style diet reduced microbial diversity and increased bacteria associated with imbalance. These changes were reflected in the immune system.
The researchers emphasize that diet plays a central role in shaping the gut ecosystem. Health guidelines recommend that adults consume about 25 to 30 grams of fiber per day, but intake in Western countries often falls short. In contrast, traditional populations may consume between 80 and 120 grams daily.
Low-fiber intake can weaken the gut microbiome, which affects not only digestion but also immunity, brain function, and mental health. Imbalances in gut bacteria have been associated with a higher risk of allergies, depression, anxiety, and neurodegenerative conditions such as Alzheimer’s disease.
Reference: “Developmental plasticity enables an intestinal tapeworm to adapt to dietary stress” by Milan Jirků, William Parker, Oldřiška Kadlecová, Martin Moos, Monika M. Wiśniewska, Roman Kuchta, Petra Tláskalová, Vladislav Ilík, Aleš Tomčala, Zuzana Pavlíčková, Kristýna Brožová, Julius Lukeš, Miroslav Oborník, Martin Kolísko, Barbora Pafčo and Kateřina Jirků, 20 February 2026, Nature Communications.
DOI: 10.1038/s41467-026-69475-0
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.