A new study helps explain why a promising class of cancer drugs has often failed to meet expectations in clinical trials.
For more than ten years, researchers have tested a group of cancer drugs known as BET inhibitors with high hopes. The science appeared solid. Many tumors rely on oncogenes that “Bromo- and Extra-Terminal domain” (BET) proteins help switch on, so blocking these proteins was expected to slow cancer growth. While this approach often worked in laboratory experiments, patient outcomes were far less encouraging, with modest benefits, notable side effects, and no reliable way to predict who would respond.
A new study from the Max Planck Institute of Immunobiology and Epigenetics (MPI-IE) in Freiburg offers insight into why these treatments have underperformed and suggests a path toward more precise therapies.
Treating an entire protein family as single target
These drugs were designed to block a shared feature that all BET proteins use to attach to chromatin, the tightly packed structure of DNA and proteins that controls gene activity. The idea was that shutting down this interaction would silence the systems that activate oncogenes. This strategy assumed that all BET proteins function in similar ways.
Research from the lab of Asifa Akhtar challenges that assumption. The team found that two major BET proteins, BRD2 and BRD4, carry out different tasks at separate stages of gene activation. BRD4 controls a later step targeted by most current treatments by releasing RNA Polymerase II, the enzyme that drives genes into active transcription. BRD2 works earlier, helping assemble and organize the molecular components needed to begin transcription.
A molecular stage manager
Drugs that block both BRD2 and BRD4 at the same time interfere with two distinct steps in the same process, leading to complex and unpredictable effects.
“Think of gene activation like stage production. BRD2 sets up the stage: assembling the props, costumes and actors to ensure preparations run smoothly. BRD2 then gives BRD4, the actor, the “start” signal to begin with the performance,” says Asifa Akhtar, who led the study at the MPI-IE. “Previous studies had been focused almost entirely on the performance. Our data shows that the setup work happening before is just as critical for gene activation,” explains Asifa Akhtar.
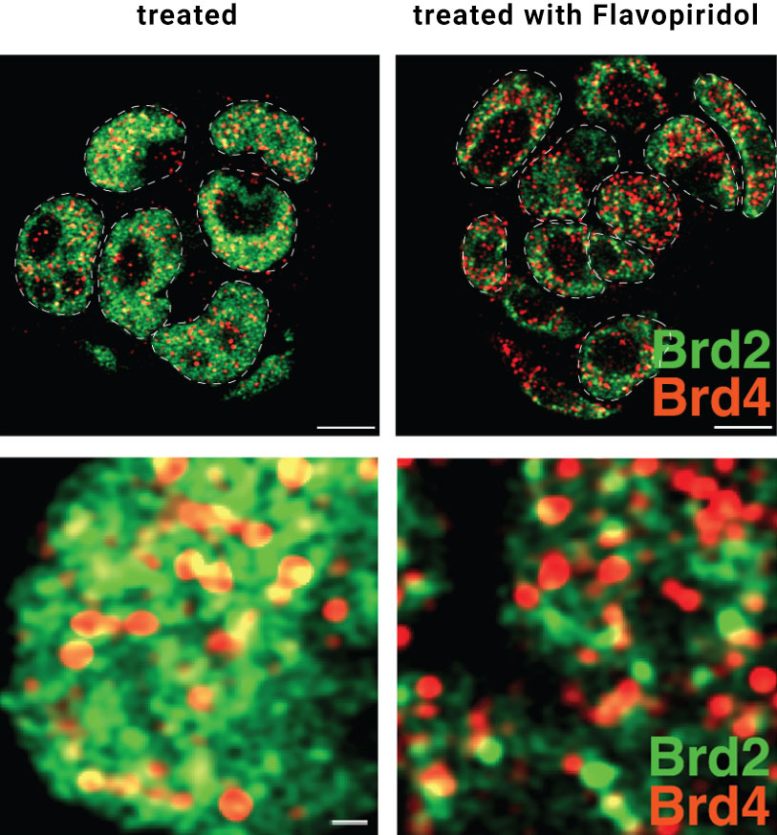
BRD2 was once viewed as the less important protein, but the new findings suggest otherwise. One reason is its sensitivity to specific chemical signals. The enzyme MOF adds markers called histone acetylations to chromatin. These act like a labeling system that helps determine which genes are read and directs BRD2 to the right locations.
BRD2 depends heavily on these bookmarks: when MOF is removed, BRD2 can no longer bind effectively to chromatin, while other BET proteins remain largely unaffected. “The findings support a model in which acetylated chromatin creates a platform that allows regulatory proteins like BRD2 to concentrate and prepare the transcription machinery for when it will be needed,” says first author Umut Erdogdu from the Akhtar lab.
The power of clustering
In addition to recognizing these signals, BRD2 organizes the transcription machinery in space by forming clusters at gene sites. These clusters gather the necessary components exactly where transcription must begin. “To understand the importance of the clustering for gene transcription, we removed only the specific part of BRD2 responsible for forming clusters while leaving the rest of the protein intact,” explains Umut Erdogdu.
The outcome was clear. Even though BRD2 remained present in the nucleus, gene transcription slowed almost as much as when the protein was entirely absent. “This demonstrates that clustering is not a side effect, but a functional feature of transcription regulation. And like a stage manager, BRD2 ensures that every performer and every piece of equipment is in place before the curtain rises,” says Asifa Akhtar.
These results reshape how scientists think about targeting BET proteins. Instead of broadly blocking their shared chromatin binding function, future treatments may focus on the distinct roles of BRD2 and BRD4. Such an approach could lead to therapies that are more precise and more predictable in their effects.
Reference: “Histone acetylation-dependent clustering of BRD2 instructs transcription dynamics” by Niyazi Umut Erdogdu, Sukanya Guhathakurta, Ronald Oellers, Maria Shvedunova, Jose A. Morin, Eric M. Patrick, Janine Seyfferth, Ward Deboutte, Alejandro Gomez-Auli, Gerhard Mittler, Ibrahim I. Cissé and Asifa Akhtar, 9 April 2026, Nature Genetics.
DOI: 10.1038/s41588-026-02533-x
The study was funded by the Max Planck Society.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.