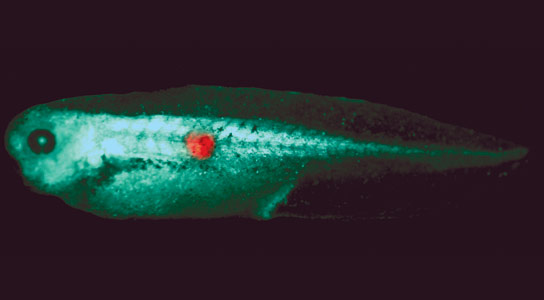
A newly published study details how researchers at Tufts University discovered a bioelectric signal that can identify cells that are likely to develop into tumors.
Biologists at Tufts University School of Arts and Sciences have discovered a bioelectric signal that can identify cells that are likely to develop into tumors. The researchers also found that they could lower the incidence of cancerous cells by manipulating the electrical charge across cells’ membranes.
“The news here is that we’ve established a bioelectric basis for the early detection of cancer,” says Brook Chernet, doctoral student and the first author of a newly published research paper co-authored with Michael Levin, Ph.D., professor of biology and director of the Center for Regenerative and Developmental Biology.
Levin notes, “We’ve shown that electric events tell the cells what to do. The voltage changes are not merely a sign of cancer. They control and direct whether the cancer occurs or not.”
Their paper, “Transmembrane Voltage Potential is an Essential Cellular Parameter for the Detection and Control of Tumor Development” will be published in the May 2013 issue of “Disease Models and Mechanisms” (available online on February 1).
Bioelectric signals underlie an important set of control mechanisms that regulate how cells grow and multiply. Chernet and Levin investigated the bioelectric properties of cells that develop into tumors in Xenopus laevis frog embryos.
In previous research, Tufts scientists have shown how manipulating membrane voltage can influence or regulate cellular behavior such as cell proliferation, migration, and shape in vivo, and be used to induce the formation or regenerative repair of whole organs and appendages. In this study, the researchers hypothesized that cancer can occur when bioelectric signaling networks are perturbed and cells stop attending to the patterning cues that orchestrate their normal development.
Tumor Cells Exhibit a Bioelectric Signature
The researchers induced tumor growth in the frog embryos by injecting the samples with mRNAs (messenger RNA) encoding well-recognized human oncogenes Gli1, KrasG12D, and Xrel3. The embryos developed tumor-like growths that are associated with human cancers such as melanoma, leukemia, lung cancer, and rhabdomyosarcoma (a soft tissue cancer that most often affects children).
When the researchers analyzed the tumor cells using a membrane voltage-sensitive dye and fluorescence microscopy, they made an exciting discovery. “The tumor sites had unique depolarized membrane voltage relative to surrounding tissue,” says Chernet. “They could be recognized by this distinctive bioelectric signal.
Changing Electrical Properties Lowers Incidence of Tumors
The Tufts biologists were also able to show that changing the bioelectric code to hyperpolarize tumor cells suppressed abnormal cell growth. “We hypothesized that the appearance of oncogene-induced tumors can be inhibited by alteration of membrane voltage,” says Levin, “and we were right.”
To counteract the tumor-inducing depolarization, they injected the cells with mRNA encoding carefully chosen ion channels (proteins that control the passage of ions across cell membranes).
Using embryos injected with oncogenes such as Xrel3, the researchers introduced one of two ion channels (the glycine gated chloride channel GlyR-F99A or the potassium channel Kir4.1) known to hyperpolarize membrane voltage gradients in frog embryos. In both cases, the incidence of subsequent tumors was substantially lower than it was with embryos that received the oncogene but no hyperpolarizing channel treatment.
Experiments to determine the cellular mechanism that allows hyperpolarization to inhibit tumor formation showed that transport of butyrate, a known tumor suppressor, was responsible
The research was supported by grants from the National Institutes of Health (awards AR061988, AR055993) and the G. Harold and Leila Y. Mathers Charitable Foundation.
Reference: “Transmembrane voltage potential is an essential cellular parameter for the detection and control of tumor development in a Xenopus model” by Brook T. Chernet and Michael Levin, 8 February 2013, Disease Models & Mechanisms.
DOI: 10.1242/dmm.010835
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.