
The study reveals how hair pigment stem cells respond to DNA damage by making fate decisions that can result in either hair graying or the development of melanoma.
Throughout life, our cells face continual challenges from both external and internal influences that can harm their DNA. This type of damage is known to play a role in aging and the development of cancer. However, scientists have long struggled to fully understand how damaged stem cells influence the long-term health and stability of tissues.
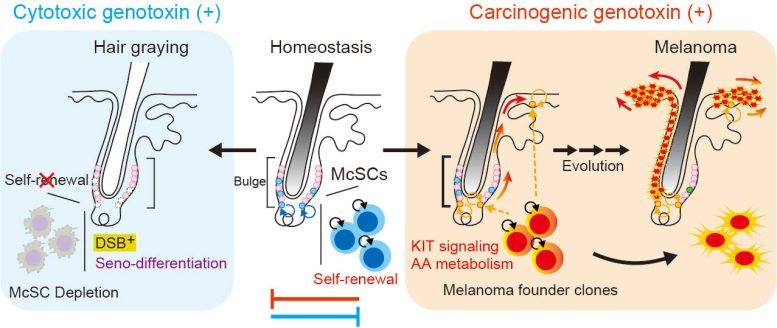
Melanocyte stem cells (McSCs) are a special type of stem cell that generates melanocytes, the pigment-producing cells responsible for hair and skin color. In mammals, these stem cells are located in the bulge–sub-bulge region of hair follicles, where they exist as immature melanoblasts. Through repeated regeneration cycles, McSCs help maintain normal pigmentation.
In a study published in Nature Cell Biology, Professor Emi Nishimura and Assistant Professor Yasuaki Mohri from The University of Tokyo explored how McSCs react to different forms of DNA damage.
Using long-term in vivo lineage tracing and gene expression profiling in mice, the researchers discovered a distinct response to DNA double-strand breaks: senescence-coupled differentiation (seno-differentiation). During this process, McSCs permanently transform into mature cells and are eventually lost, leading to hair graying. This protective mechanism is controlled by activation of the p53–p21 pathway.
When Protection Fails: The Cancer Connection
In contrast, when exposed to certain carcinogens, such as 7,12-dimethylbenz(a)anthracene or ultraviolet B, McSCs bypass this protective differentiation program—even in the presence of DNA damage. Instead, they retain self-renewal capacity and expand clonally, a process supported by KIT ligand secreted both from the local niche and within the epidermis. This niche-derived signal suppresses seno-differentiation, tipping McSCs toward a tumor-prone fate.
Nishimura says, “These findings reveal that the same stem cell population can follow antagonistic fates—exhaustion or expansion—depending on the type of stress and microenvironmental signals.” She adds, “It reframes hair graying and melanoma not as unrelated events, but as divergent outcomes of stem cell stress responses.”
Importantly, this study does not suggest that graying hair prevents cancer, but rather that seno-differentiation represents a stress-induced protective pathway that removes potentially harmful cells. Conversely, when this mechanism is bypassed, the persistence of damaged McSCs may predispose to melanomagenesis.
By identifying the molecular circuits that govern this fate bifurcation, the study provides a conceptual framework that links tissue aging and cancer, and highlights the beneficial role of eliminating potentially harmful stem cells through natural “senolysis,” resulting in a phenotype that safeguards against cancer.
Reference: “Antagonistic stem cell fates under stress govern decisions between hair greying and melanoma” by Yasuaki Mohri, Jialiang Nie, Hironobu Morinaga, Tomoki Kato, Takahiro Aoto, Takashi Yamanashi, Daisuke Nanba, Hiroyuki Matsumura, Sakura Kirino, Kouji Kobiyama, Ken J. Ishii, Masahiro Hayashi, Tamio Suzuki, Takeshi Namiki, Jun Seita and Emi K. Nishimura, 6 October 2025, Nature Cell Biology.
DOI: 10.1038/s41556-025-01769-9
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.