New research exposes the molecular switches that let some cancers shapeshift and reveals how they might finally be stopped.
Certain cancers are extremely difficult to treat, and carcinomas are among the most stubborn. Unlike many other malignancies, these tumors can behave in unusual ways. Some have the ability to shift their identity, taking on characteristics of cells from entirely different organs, including skin. This shape-changing behavior makes them especially hard to target with current therapies. “The tumors are notoriously plastic in their cellular identity,” says Cold Spring Harbor Laboratory (CSHL) Professor Christopher Vakoc. In some cases, this flexibility allows tumors to adapt and survive treatment.
New Studies Reveal Vulnerabilities in Pancreatic and Lung Cancer
Recent research from the Vakoc lab has uncovered important weaknesses in two particularly challenging carcinomas. According to Vakoc, these discoveries may “tee up targets for therapy.”
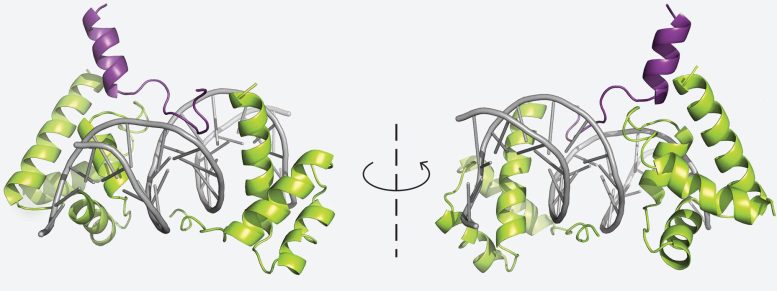
In a study published in Nature Communications, CSHL scientists identified a protein that controls whether pancreatic cancer cells keep their traditional identity or begin to resemble and behave like skin cells. In separate work reported in Cell Reports, the team determined the crystal structure of another group of proteins that is central to tuft cell lung cancer.
From Discovery to Potential Epigenetic Therapy
Vakoc describes this progress as a full-circle moment for the laboratory. When tuft cell lung cancer was first identified in 2018, he and his colleagues were searching for epigenetic drivers of tumor growth. Their focus extended beyond DNA sequences to the mechanisms that regulate gene activity, including transcription and gene regulation. Now, working with CSHL Director of Research Leemor Joshua-Tor, the group has identified findings that could eventually form the basis of an epigenetic therapy aimed at stopping tumor growth.
Master Regulators of Cellular Identity
These studies advance a long-term objective that has guided Vakoc’s research for 17 years. “We aim to identify the master regulators of cellular identity,” he says. The ultimate goal is to develop therapies that target these “master regulators,” similar to how hormone treatments transformed care for breast and prostate cancers that were once far more difficult to manage. Still, Vakoc emphasizes that translating these insights into medicines will take time.
Toward More Precise and Safer Cancer Treatments
If new drugs emerge from this work, Vakoc hopes they will attack cancer cells without harming healthy tissues. That commitment to precision is reflected in the lab’s recent experiments. In mouse models of pancreatic and lung cancer, the researchers observed no signs of toxicity or injury to major organs. “We’re setting a higher bar for specificity when it comes to new cancer targets and treatments,” Vakoc says.
Beyond the search for future therapies, the team is also deepening scientific understanding of how cellular identity is controlled. That growing knowledge could help shape a more precise and effective standard of care in the years ahead.
Reference: “Structural basis of DNA-dependent coactivator recruitment by the tuft cell master regulator POU2F3” by Aktan Alpsoy, Jonathan J. Ipsaro, Damianos Skopelitis, Sujay Pal, Frank S. Chung, Shannon Carpenter, John J. Desmarais, Xiaoli S. Wu, Kenneth Chang, Matthew T. DiMare, Erin Harten, Staci Bergman, Justin B. Kinney, Jeffrey A. Engelman, Hyo-Eun C. Bhang, Leemor Joshua-Tor and Christopher R. Vakoc, 18 November 2025, Cell Reports.
DOI: 10.1016/j.celrep.2025.116572
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Wouldn’t it be a greater achievement to ultimately and safely cure all cancers before we all pass-on!!!!!!!❤️🙏🏻🇺🇸👍🏼