Researchers at the University of Michigan Life Sciences Institute and the Howard Hughes Medical Institute have determined how satellite DNA, considered to be “junk DNA,” plays a crucial role in holding the genome together.
Their findings, published recently in the journal eLife, indicate that this genetic “junk” performs the vital function of ensuring that chromosomes bundle correctly inside the cell’s nucleus, which is necessary for cell survival. And this function appears to be conserved across many species.
This pericentromeric satellite DNA consists of a very simple, highly repetitive sequence of genetic code. Although it accounts for a substantial portion of our genome, satellite DNA does not contain instructions for making any specific proteins. What’s more, its repetitive nature is thought to make the genome less stable and more susceptible to damage or disease. Until fairly recently, scientists believed this so-called “junk” or “selfish” DNA did not serve any real purpose.
“But we were not quite convinced by the idea that this is just genomic junk,” said Yukiko Yamashita, research professor at the LSI and lead author on the study. “If we don’t actively need it, and if not having it would give us an advantage, then evolution probably would have gotten rid of it. But that hasn’t happened.”
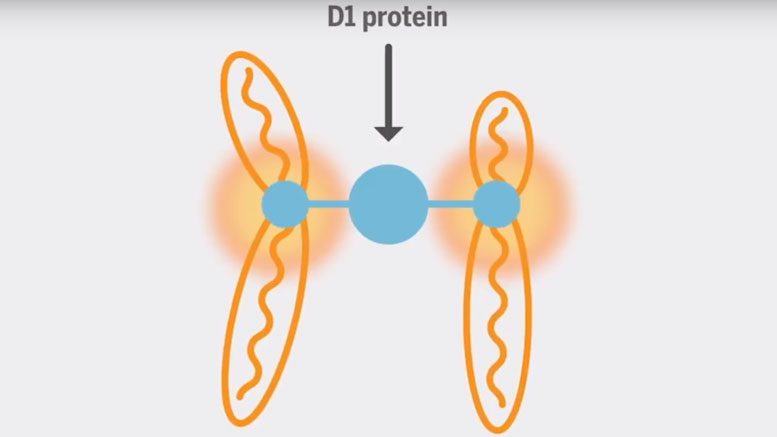
Yamashita and her colleagues decided to see what would happen if cells could not use this pericentromeric satellite DNA. Because it exists in long, repetitive sequences, the researchers could not simply mutate or cut the entire satellite DNA out of the genome. Instead, they approached the question through D1, a protein known to bind to satellite DNA.
The researchers removed D1 from the cells of a commonly used model organism, Drosophila melanogaster (fruit flies). And the team quickly noticed that germ cells—the cells that ultimately develop into sperm or eggs—were dying.
Further analysis revealed that the dying cells were forming micro-nuclei, or tiny buds, outside the nucleus that included pieces of the genome. Without the entire genome encapsulated in the nucleus, the cells could not survive.
The researchers believe that the D1 protein binds onto the satellite DNA to pull all of the chromosomes together in the nucleus. If the D1 protein cannot grab the satellite DNA, the cell loses its ability to form a complete nucleus and ultimately dies.
“It’s like forming a bouquet,” said Yamashita, who is also a professor of cell and developmental biology at the U-M Medical School and an HHMI investigator. “The protein has multiple binding sites, so it can bind onto multiple chromosomes and package them together in one place, preventing individual chromosomes from floating out of the nucleus.”
The team conducted similar experiments using mouse cells and found the same results: When they removed a protein that normally binds to mouse satellite DNA, the cells again formed micro-nuclei and did not survive.
The similar findings from both fruit fly and mouse cells lead Yamashita and her colleagues to believe that satellite DNA is essential for cellular survival, not just in model organisms, but across species that embed DNA into the nucleus—including humans.
Reference: “A conserved function for pericentromeric satellite DNA” by Madhav Jagannathan, Ryan Cummings, Yukiko M Yamashita, 16 March 2018, eLife.
DOI: 10.7554/eLife.34122
The research was supported by the Howard Hughes Medical Institute, the National Institutes of Health, and the American Heart Association.
In addition to Yamashita, other authors of the study are Madhav Jagannathan and Ryan Cummings, also of U-M.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.