
Researchers are developing coatings for electronic components to integrate them more easily into the body for tumor monitoring and tissue replacement.
Although true “cyborgs” — part human, part robotic beings — are science fiction, researchers are taking steps toward integrating electronics with the body. Such devices could monitor for tumor development or stand in for damaged tissues. But connecting electronics directly to human tissues in the body is a huge challenge. Now, a team is reporting new coatings for components that could help them more easily fit into this environment.
The researchers will present their results today (August 17, 2020) at the American Chemical Society (ACS) Fall 2020 Virtual Meeting & Expo. ACS is holding the meeting through Thursday. It features more than 6,000 presentations on a wide range of science topics.
“We got the idea for this project because we were trying to interface rigid, inorganic microelectrodes with the brain, but brains are made out of organic, salty, live materials,” says David Martin, Ph.D., who led the study. “It wasn’t working well, so we thought there must be a better way.”
Traditional microelectronic materials, such as silicon, gold, stainless steel, and iridium, cause scarring when implanted. For applications in muscle or brain tissue, electrical signals need to flow for them to operate properly, but scars interrupt this activity. The researchers reasoned that a coating could help.
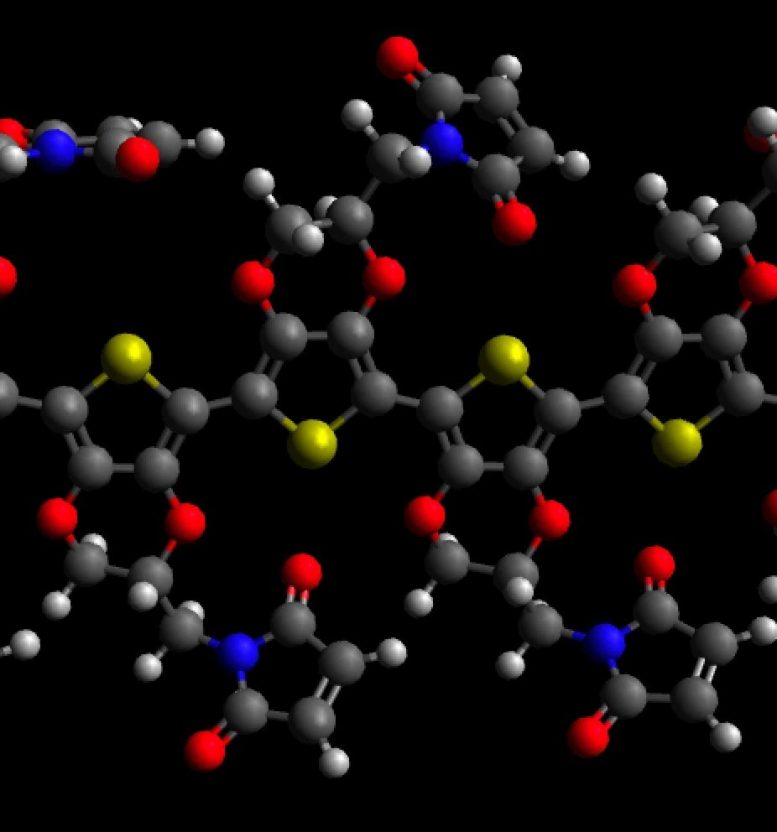
Molecular model of PEDOT with maleimide; carbon atoms are grey, oxygens red, nitrogens blue, sulfurs yellow and hydrogens white. Credit: David Martin
“We started looking at organic electronic materials like conjugated polymers that were being used in non-biological devices,” says Martin, who is at the University of Delaware. “We found a chemically stable example that was sold commercially as an antistatic coating for electronic displays.” After testing, the researchers found that the polymer had the properties necessary for interfacing hardware and human tissue.
“These conjugated polymers are electrically active, but they are also ionically active,” Martin says. “Counter ions give them the charge they need so when they are in operation, both electrons and ions are moving around.” The polymer, known as poly(3,4-ethylenedioxythiophene) or PEDOT, dramatically improved the performance of medical implants by lowering their impedance two to three orders of magnitude, thus increasing signal quality and battery lifetime in patients.
Martin has since determined how to specialize the polymer, putting different functional groups on PEDOT. Adding a carboxylic acid, aldehyde or maleimide substituent to the ethylenedioxythiophene (EDOT) monomer gives the researchers the versatility to create polymers with a variety of functions.
“The maleimide is particularly powerful because we can do click chemistry substitutions to make functionalized polymers and biopolymers,” Martin says. Mixing unsubstituted monomer with the maleimide-substituted version results in a material with many locations where the team can attach peptides, antibodies or DNA. “Name your favorite biomolecule, and you can in principle make a PEDOT film that has whatever biofunctional group you might be interested in,” he says.
Most recently, Martin’s group created a PEDOT film with an antibody for vascular endothelial growth factor (VEGF) attached. VEGF stimulates blood vessel growth after injury, and tumors hijack this protein to increase their blood supply. The polymer that the team developed could act as a sensor to detect overexpression of VEGF and thus early stages of disease, among other potential applications.
Other functionalized polymers have neurotransmitters on them, and these films could help sense or treat brain or nervous system disorders. So far, the team has made a polymer with dopamine, which plays a role in addictive behaviors, as well as dopamine-functionalized variants of the EDOT monomer. Martin says these biological-synthetic hybrid materials might someday be useful in merging artificial intelligence with the human brain.
Ultimately, Martin says, his dream is to be able to tailor how these materials deposit on a surface and then put them in tissue in a living organism. “The ability to do the polymerization in a controlled way inside a living organism would be fascinating.”
A press conference on this topic will be held Wednesday, August 19, 2020, at 10 a.m. Eastern time online.
The researchers acknowledge support and funding from the National Science Foundation and the University of Delaware.





I’d rather die.
Wow how cyberpunk! I’m s#!tting and Cumming!
What’s with all the cumming and going?
I’m holding out for a neural link myself. Or at least implanted AR lenses.