Hormone-regulated immune cells produce IL-10 to resolve pain more effectively in males, offering a potential new target for non-opioid chronic pain treatments.
Chronic pain often lingers longer in women than in men. A new study suggests that differences in immune cells known as monocytes could be a key reason. These cells are regulated in part by sex hormones.
In research published in Science Immunology, scientists at Michigan State University identified a specific group of monocytes that release a pain-reducing molecule. The team found that these cells are more active in males, likely because of higher levels of sex hormones such as testosterone.
In females, monocytes produced lower levels of this pain-dampening signal, which was linked to prolonged pain and slower recovery. Geoffroy Laumet, MSU associate professor of physiology, and Jaewon Sim, a former graduate student in his lab, observed the same pattern in both mouse models and human patients.
The work, supported by the National Institutes of Health and the Department of Defense, suggests it may eventually be possible to encourage these immune cells to generate stronger pain-calming signals. Although practical treatments are likely many years away, Laumet believes the findings could ultimately lead to non-opioid options that provide relief for millions of people while also reinforcing that women’s pain has a clear biological basis.

“The difference in pain between men and women has a biological basis,” Laumet said. “It’s not in your head, and you’re not soft. It’s in your immune system.”
Chronic Pain Perception and Diagnostic Challenges
Pain begins when neurons throughout the body respond to stimulation. Under normal conditions, these nerve cells remain quiet until triggered by an injury such as stubbing a toe or falling off a bike. In people with chronic pain, however, these sensors can become active with very mild stimulation or even without any obvious trigger.
Clinicians still depend heavily on self-reported pain scores, usually asking patients to rate discomfort on a scale from one to 10. Because pain is highly personal and subjective, differences in reporting can complicate diagnosis. When women report chronic or long-lasting pain more often than men, those differences are sometimes attributed to perception rather than biology.
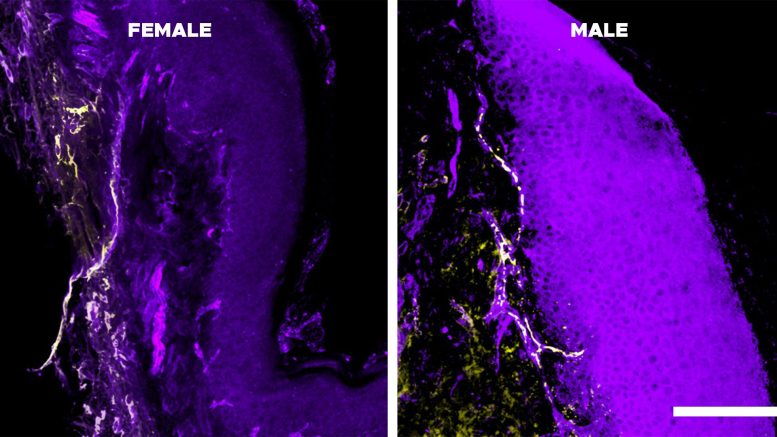
Laumet has focused his laboratory on understanding pain for the past six years. During a small pilot study, his team noticed that males showed higher levels of interleukin-10, or IL-10. A second round of testing confirmed elevated levels of this molecule, which signals neurons to quiet pain responses, strengthening their confidence that the pattern was meaningful.
“That was the turning point for me,” Sim said. “I feel extremely fortunate that we trusted those early, uncertain findings and chose to pursue them further.”
IL-10, Immune-Neural Communication, and Active Pain Resolution
To investigate further, the researchers used high-dimensional spectral flow cytometry, an advanced method for analyzing immune cells. They discovered that monocytes, once considered mainly precursor cells with limited direct function, actively communicate with pain-sensing neurons by releasing IL-10. The team found that IL-10-producing monocytes were significantly more active in males than in females. When male sex hormones were blocked, this effect reversed.
“This study shows that pain resolution is not a passive process,” Laumet said. “It is an active, immune-driven one.”

The researchers conducted at least five different types of experiments in mouse models to confirm their observations. Each approach produced consistent results.
Laumet then collaborated with Sarah Linnsteadt at the University of North Carolina at Chapel Hill, who studies psychological outcomes in people involved in car accidents. Her data revealed a similar trend: men had more active IL-10-producing monocytes and recovered from pain more quickly.
Toward Non-Opioid Therapies Targeting IL-10 Pathways
Together, these findings highlight an immune–neural pathway that influences how pain subsides, shifting attention from the triggers of pain to the mechanisms that allow it to persist. Future research will explore whether treatments can safely enhance IL-10 production along this pathway. Instead of simply blocking pain signals, such therapies could help the body resolve pain more effectively.
“Future researchers can build on this work,” Laumet said. “This opens new avenues for non-opioid therapies aimed at preventing chronic pain before it’s established.”
Reference: “Monocyte-derived IL-10 drives sex differences in pain duration” by Jaewon Sim, Elizabeth O’Guin, Chiho Sugimoto, Sophie Laumet, Karli Monahan, Matthew P. Bernard, Samuel A. McLean, Liz M. Albertorio-Sáez, Ying Zhao, Hariharan Ramakrishnan, Sabrina de Souza, Olivia C. Eller, Christine J. Smoyer, Kyle M. Baumbauer, Matthias Mack, Joseph K. Folger, Alfred J. Robison, Sarah D. Linnstaedt and Geoffroy Laumet, 20 February 2026, Science Immunology.
DOI: 10.1126/sciimmunol.adx0292
This study was funded by NIH/National Institutes of Health and the U.S. Department of Defense.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
I suspect there is more to this story than just IL-10 sex differences. During an interesting phase of my life more than 45 years ago I observed that the loss of most male sex hormones and strong increase in female hormones my sensitivity to pain dramatically changed. The pain was both more intensely felt and easier to tolerate. This might make a nice but difficult study for people in this field. The precise path from where I was to where I am now may hold explanations why I handle and tolerate pain “oddly”. I can get up and walk when doctors think I should be curled up in bed crying. (But when a girl has to go and hates the thought of a wet bed, she just does what she must. Is this pain perception or pain handling? I don’t know. I’d like to, though.)
{^_^}