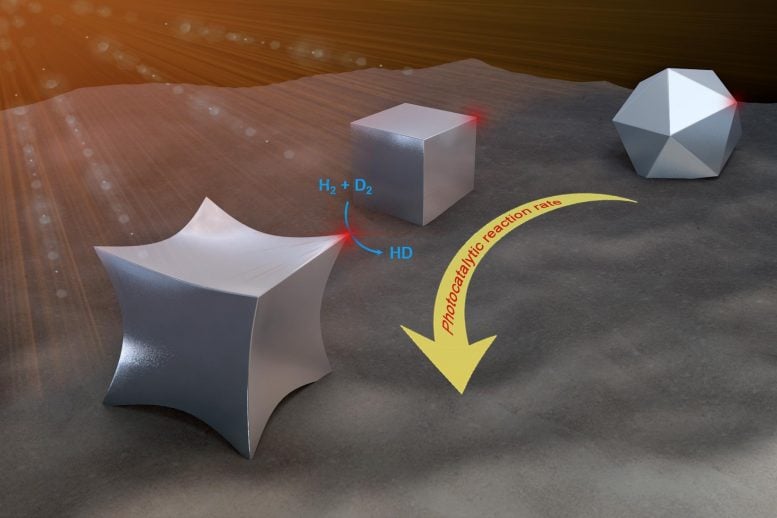
Study: Pointed tips on aluminum ‘octopods’ increase catalytic reactivity.
Points matter when designing nanoparticles that drive important chemical reactions using the power of light.
Researchers at Rice University’s Laboratory for Nanophotonics (LANP) have long known that a nanoparticle’s shape affects how it interacts with light, and their latest study shows how shape affects a particle’s ability to use light to catalyze important chemical reactions.
In a comparative study, LANP graduate students Lin Yuan and Minhan Lou and their colleagues studied aluminum nanoparticles with identical optical properties but different shapes. The most rounded had 14 sides and 24 blunt points. Another was cube-shaped, with six sides and eight 90-degree corners. The third, which the team dubbed “octopod,” also had six sides, but each of its eight corners ended in a pointed tip.

All three varieties have the ability to capture energy from light and release it periodically in the form of super-energetic hot electrons that can speed up catalytic reactions. Yuan, a chemist in the research group of LANP director Naomi Halas, conducted experiments to see how well each of the particles performed as photocatalysts for hydrogen dissociation reaction. The tests showed octopods had a 10 times higher reaction rate than the 14-sided nanocrystals and five times higher than the nanocubes. Octopods also had a lower apparent activation energy, about 45% lower than nanocubes and 49% lower than nanocrystals.

“The experiments demonstrated that sharper corners increased efficiencies,” said Yuan, co-lead author of the study, which is published in the American Chemical Society journal ACS Nano. “For the octopods, the angle of the corners is about 60 degrees, compared to 90 degrees for the cubes and more rounded points on the nanocrystals. So the smaller the angle, the greater the increase in reaction efficiencies. But how small the angle can be is limited by chemical synthesis. These are single crystals that prefer certain structures. You cannot make infinitely more sharpness.”
Lou, a physicist and study co-lead author in the research group of LANP’s Peter Nordlander, verified the results of the catalytic experiments by developing a theoretical model of the hot electron energy transfer process between the light-activated aluminum nanoparticles and hydrogen molecules.
“We input the wavelength of light and particle shape,” Lou said. “Using these two aspects, we can accurately predict which shape will produce the best catalyst.”
The work is part of an ongoing green chemistry effort by LANP to develop commercially viable light-activated nanocatalysts that can insert energy into chemical reactions with surgical precision. LANP has previously demonstrated catalysts for ethylene and syngas production, the splitting of ammonia to produce hydrogen fuel, and for breaking apart “forever chemicals.”
“This study shows that photocatalyst shape is another design element engineers can use to create photocatalysts with higher reaction rates and lower activation barriers,” said Halas, Rice’s Stanley C. Moore Professor of Electrical and Computer Engineering, director of Rice’s Smalley-Curl Institute and a professor of chemistry, bioengineering, physics and astronomy, and materials science and nanoengineering.
Reference: “Morphology-Dependent Reactivity of a Plasmonic Photocatalyst” by Lin Yuan, Minhan Lou, Benjamin D. Clark, Minghe Lou, Linan Zhou, Shu Tian, Christian R. Jacobson, Peter Nordlander and Naomi J. Halas, 12 August 2020, ACS Nano.
DOI: 10.1021/acsnano.0c05383
Nordlander is the Wiess Chair and Professor of Physics and Astronomy, and professor of electrical and computer engineering, and materials science and nanoengineering.
Additional study co-authors include Benjamin Clark, Minghe Lou, Linan Zhou, Shu Tian and Christian Jacobson, all of Rice. The research was supported by the Welch Foundation (C-1220, C-1222), the Air Force Office of Scientific Research (FA9550-15-1-0022), the Defense Threat Reduction Agency (HDTRA 1-16-1-0042) and the Department of Defense National Defense Science & Engineering Graduate Fellowship Program.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.