
The body’s natural pain killers can be enhanced, offering a potential alternative with fewer side effects compared to opioids.
A study in cells and mice finds compound works with fewer side effects than opioids.
Fentanyl, oxycodone, morphine—these substances are familiar to many as a source of both pain relief and the cause of a painful epidemic of addiction and death.
Scientists have attempted for years to balance the potent pain-relieving properties of opioids with their numerous negative side effects—with mostly mixed results.
Work by John Traynor, Ph.D., and Andrew Alt, Ph.D., and their team at the University of Michigan Edward F. Domino Research Center, funded by the National Institute on Drug Abuse, seeks to side-step these problems by harnessing the body’s own ability to block pain.
All opioid drugs—from poppy-derived opium to heroin—work on receptors that are naturally present in the brain and elsewhere in the body. One such receptor, the mu-opioid receptor, binds to natural pain-killers in the body called endogenous endorphins and enkephalins. Drugs acting on the mu-opioid receptor can cause addiction as well as unwanted side effects like drowsiness, problems with breathing, constipation, and nausea.
“Normally, when you are in pain, you are releasing endogenous opioids, but they’re just not strong enough or long lasting enough,” says Traynor. The team had long hypothesized that substances called positive allosteric modulators could be used to enhance the body’s own endorphins and enkephalins. In a new paper published in PNAS, they demonstrate that a positive allosteric modulator known as BMS-986122 can boost enkephalins’ ability to activate the mu-opioid receptor.
What’s more, unlike opioid drugs, positive allosteric modulators only work in the presence of endorphins or enkephalins, meaning they would only kick in when needed for pain relief. They do not bind to the receptor in the way that opioids do instead binding in a different location that enhances their ability to respond to the body’s pain-relieving compounds.
“When you need enkephalins, you release them in a pulsatile fashion in specific regions of the body, then they are metabolized quickly,” explains Traynor. “In contrast, a drug like morphine floods the body and brain and sticks around for several hours.”
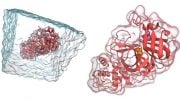
The team demonstrated the modulator’s ability to stimulate the mu-opioid receptor by isolating the purified receptor and measuring how it responds to enkephalins. “If you add the positive allosteric modulator, you need a lot less enkephalin to get the response.”
Additional electrophysiology and mouse experiments confirmed that the opioid receptor was more strongly activated by the body’s pain-relieving molecules leading to pain relief. In contrast, the modulator showed much reduced side effects of depression of breathing, constipation, and addiction liability.
Their next goal is to measure their ability to enhance activation of endogenous opioids under conditions of stress or chronic pain, explains Traynor, to ensure that they are effective but don’t lead to more dangerous responses like depression of breathing.
“While these molecules won’t solve the opioid crisis,” says Traynor, “they could slow it and prevent it from happening again because patients in pain could take this type of a drug instead of a traditional opioid drug.”
Reference: “Positive allosteric modulation of the mu-opioid receptor produces analgesia with reduced side effects” by Ram Kandasamy, Todd M. Hillhouse, Kathryn E. Livingston, Kelsey E. Kochan, Claire Meurice, Shainnel O. Eans, Ming-Hua Li, Andrew D. White, Bernard P. Roques, Jay P. McLaughlin, Susan L. Ingram, Neil T. Burford, Andrew Alt and John R. Traynor, 20 April 2021, Proceedings of National Academy of Sciences.
DOI: 10.1073/pnas.2000017118
Funding: National Institutes of Health







Good to know somebody is working on some new approaches. Basically it has appeared that scientist and medical professional have turned their back on those in need. The so called war on opioids has been a miserable failure. You find now that physicians literally hate their chronic pain patients and are now are impowered to just sh*t on them. Isolated and misrepresented we withdraw and self medicate. With all your brilliance your you seem to miss simple points.
There was never a problem war to prevent addiction to opioids. I don’t even recall seeing an attempt to fake one. Big pharm lines a lot of pockets and Purdue Pharma walked away billionaires after their actions to increase prescriptions of oxytocin despite being aware of the addiction and growing problem that they call the opioid crisis. Then the US government agreed to take the company instead of the massive fine the Slacker family says they can’t pay. Purdue pharmacy along with many others lobbied so hard against what the DEA was doing to curb their actions that eventually legislation was passed that tied their hands so they can no long pursue big pharm companies or hold them to any real consequence. Doctors and nurses are so propagandized they regurgitate “normal” to addicts like they are weak or less than human if they take opioid medication other than prescribed or it doesn’t control their pain. Tolerance is real. If you have a patient who has ever needed an opioid prescription increased they do not take them as prescribed but they have a ritual that gets them from month to and might even involve purchasing other opioid illegally as they have to take everything so the medical community does strip their prescriptions away all together..Now the day no one renews your scripts is either the first day of freedom or the beginning of the end depends on the person. I am one of a few who want to know how your taking it in reality cause that information is helpful on several levels but I won’t even tell others I want to know how they really take their prescriptions because if I’m not sitting in judgment on them I must understand because I’m an addict myself. The whole circle of bullsh*t needs to end. I understand because I work intensive care for 20 years and talked to many survivors or accidental or intentional overdoses, not to mention, tens of thousands of addicts whose tolerance is important to controlling acute pain or if they are withdrawling because I’m under dosing them.
Hello, just giving you some feedback. I am a retired RN, disabled. I have chronic pain from Back problems and significant PMR. I have run the gamete. 8 different procedures on my back. I also have advanced CAD. I am on opioids because I can not use Motrin ect. In short my physician has taken mercy on me and remains supportive. The rest of the medical community treats me with distain. I feel trapped and alone. I would self medicate in a heart beat. I am trying to survive. If the pain gets out of control I go into complete despair. My tolerance is up there but there is no way I would ask for more from them, I am already pushing it with them. Why has the medical community turned its back on me?
Hopefully it will work to get people off of opioids.
The issue of addiction has politicized and stigmatized the use of opiotes for patients. Stories of promising future treatments are just that until they are proven effective in the real world. Medical and scientific journals might consider toning down the rhetoric about addiction which seems to insert itself in every story. It is time to disentangle addiction issues from a clear need to treat pain.