
The new device fits around an Ambu bag (blue), which hospitals already have on hand in abundance. Designed to be squeezed by hand, instead they are squeezed by mechanical paddles (center) driven by a small motor. This directs air through a tube which is placed in the patient’s airway. Credit: Courtesy of the researchers
Clinical and design considerations will be published online; goal is to support rapid scale-up of device production to alleviate hospital shortages.
One of the most pressing shortages facing hospitals during the Covid-19 emergency is a lack of ventilators. These machines can keep patients breathing when they no longer can on their own, and they can cost around $30,000 each. Now, a rapidly assembled volunteer team of engineers, physicians, computer scientists, and others, centered at MIT, is working to implement a safe, inexpensive alternative for emergency use, which could be built quickly around the world.
The team, called MIT E-Vent (for emergency ventilator), was formed on March 12, 2020, in response to the rapid spread of the Covid-19 pandemic. Its members were brought together by the exhortations of doctors, friends, and a sudden flood of mail referencing a project done a decade ago in the MIT class 2.75 (Medical Device Design). Students working in consultation with local physicians designed a simple ventilator device that could be built with about $100 worth of parts. They published a paper detailing their design and testing, but the work ended at that point. Now, with a significant global need looming, a new team, linked to that course, has resumed the project at a highly accelerated pace.
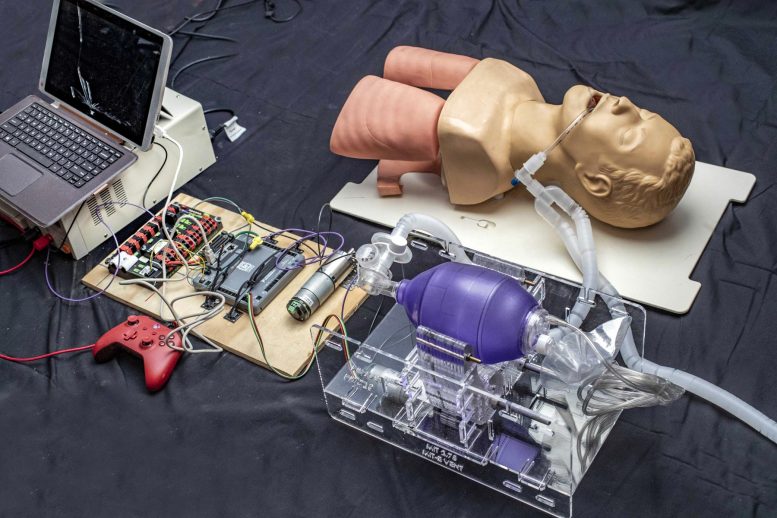
This shows the setup used for preliminary testing of an earlier version of the low-cost prototype design that could provide rapid deployment to hospitals facing shortages of the vital equipment. Credit: Courtesy of the researchers
The key to the simple, inexpensive ventilator alternative is a hand-operated plastic pouch called a bag-valve resuscitator, or Ambu bag, which hospitals already have on hand in large quantities. These are designed to be operated by hand, by a medical professional or emergency technician, to provide breaths to a patient in situations like cardiac arrest, until an intervention such as a ventilator becomes available. A tube is inserted into the patient’s airway, as with a hospital ventilator, but then the pumping of air into the lungs is done by squeezing and releasing the flexible pouch. This is a task for skilled personnel, trained in how to evaluate the patient and adjust the timing and pressure of the pumping accordingly.
The innovation begun by the earlier MIT class, and now being rapidly refined and tested by the new team, was to devise a mechanical system to do the squeezing and releasing of the Ambu bag, since this is not something that a person could be expected to do for any extended period. But it is crucial for such a system to not damage the bag and to be controllable, so that the amount of air and pressures being delivered can be tailored to the particular patient. The device must be very reliable, since an unexpected failure of the device could be fatal, but as designed by the MIT team, the bag can be immediately operated manually.
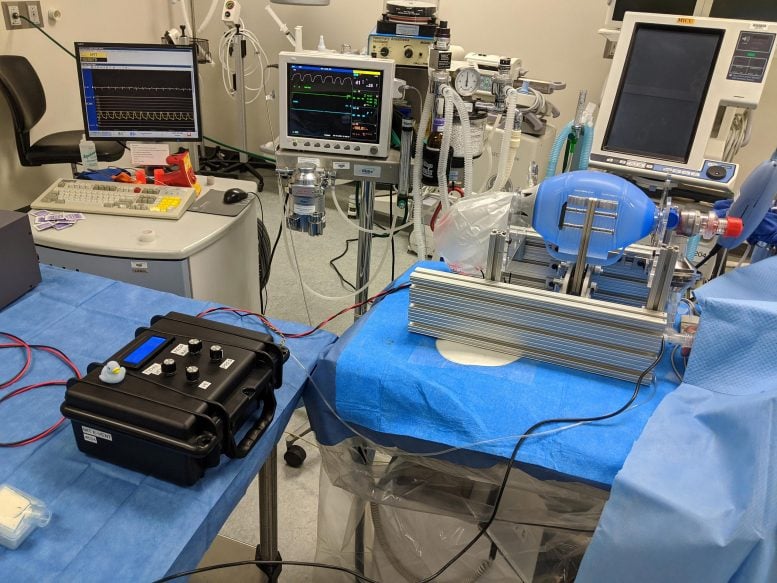
Test setup in the lab shows the most recent version of the device undergoing initial testing. Credit: Courtesy of the researchers
The team is particularly concerned about the potential for well-meaning but inexperienced do-it-yourselfers to try to reproduce such a system without the necessary clinical knowledge or expertise with hardware that can operate for days; around 1 million cycles would be required to support a ventilated patient over a two-week period. Furthermore, it requires code that is fault-tolerant, since ventilators are precision devices that perform a life-critical function. To help curtail the spread of misinformation or poorly-thought-out advice, the team has added to their website verified information resources on the clinical use of ventilators and the requirements for training and monitoring in using such systems. All of this information is freely available at e-vent.mit.edu.
“We are releasing design guidance (clinical, mechanical, electrical/controls, testing) on a rolling basis as it is developed and documented,” one team member says. “We encourage capable clinical-engineering teams to work with their local resources, while following the main specs and safety information, and we welcome any input other teams may have.”
The researchers emphasize that this is not a project for typical do-it-yourselfers to undertake, since it requires specialized understanding of the clinical-technical interface, and the ability to work in consideration of strict U.S. Food and Drug Administration specifications and guidelines.

Close up of one version of the prototype shows how an emergency resuscitation (Ambu) bag, in purple, fits into the device. Credit: Courtesy of the researchers
Such devices “have to be manufactured according to FDA requirements, and should only be utilized under the supervision of a clinician,” a team member said. “The Department of Health and Human Services released a notice stating that all medical interventions related to Covid-19 are no longer subject to liability, but that does not change our burden of care.” he said. “At present, we are awaiting FDA feedback” about the project. “Ultimately, our intent is to seek FDA approval. That process takes time, however.”
The all-volunteer team is working without funding and operating anonymously for now because many of them have already been swamped by inquiries from people wanting more information, and are concerned about being overwhelmed by calls that would interfere with their work on the project. “We would really, really like to just stay focused,” says one team member. “And that’s one of the reasons why the website is so essential, so that we can communicate with anyone who wants to read about what we are doing, and also so that others across the world can communicate with us.”
“The primary consideration is patient safety. So we had to establish what we’re calling minimum clinical functional requirements,” that is, the minimum set of functions that the device would need to perform to be both safe and useful, says one of the team members, who is both an engineer and an MD. He says one of his jobs is to translate between the specialized languages used by the engineers and the medical professionals on the team.
That determination of minimum requirements was made by a team of physicians with broad clinical backgrounds, including anesthesia and critical care, he says. In parallel, the group set to work on designing, building, and testing an updated prototype. Initial tests revealed the high loads that actual use incurs, and some weaknesses that have already been addressed so that, in the words of team co-leads, “Even the professor can kick it across the room.” In other words, early attempts focused on super “makability” were too optimistic.
New versions have already been fabricated and are being prepared for additional functional tests. Already, the team says there is enough detailed information on their website to allow other teams to work in parallel with them, and they have also included links to other teams that are working on similar design efforts.
In under a week the team has gone from empty benches to their first realistic tests of a prototype. One team member says that in the less than a week full they have been working, motivated by reports of doctors already having to ration ventilators, and the intense focus the diverse group has brought to this project, they have already generated “multiple theses worth” of research.
The cross-disciplinary nature of the group has been crucial, one team member says. “The most exciting times and when the team is really moving fast are when we have an a design engineer, sitting next to a controls engineer, sitting next to the fabrication expert, with an anesthesiologist on WebEx, all solid modeling, coding, and spreadsheeting in parallel. We are discussing the details of everything from ways to track patients’ vital signs data to the best sources for small electric motors.”
The intensity of the work, with people putting in very long hours every day, has been tiring but hasn’t dulled their enthusiasm. “We all work together, and ultimately the goal is to help people, because people’s lives understandably hang in the balance,” he said.
Reference: “Design and Prototyping of a Low-cost Portable Mechanical Ventilator” by Abdul Mohsen Al Husseini, Heon Ju Lee, Justin Negrete, Stephen Powelson, Amelia Servi, Alexander Slocum and Jussi Saukkonen.
PDF
Update: MIT Team Races to Fill COVID-19 Ventilator Shortage With Low-Cost, Open-Source Alternative.







Where are the plans? The PDF reference on this page is to a 2010 paper. The https://e-vent.mit.edu/ page makes reference to plans, but no plans are apparent. I have a machine shop idled in Illinois and the owner wants to help. This is within their capability.
“Clinical and design considerations will be published online”
Where are the plans? The PDF reference on this page is to a 2010 paper. The https://e-vent.mit.edu/ page makes reference to plans, but no plans are apparent. How can we help if you post false info like this ,,, shame
These people are trying to help, and attacking them doesn’t help the situation we are all in together.
“Clinical and design considerations will be published online”
We have a different design… Care to work with us as well?
https://youtu.be/uV9nWFrlbmI
How about durability of the ambu bag itself – the pouch and the valves?
Need the plans.
https://e-vent.mit.edu/
Wouldn’t it have made more sense to reengineer an approved standard pneumatic ventilator like the Bird mk. 7 or 8 for ease of manufacture, rather than use a (single use) Ambubag (with valves designed for relatively short use, hours, perhaps a day)?
New name: “Bag’em Bot”!
Of course it’s going to work. It’s something anyone with an Ambu bag (or equivalent) an Arduino, some tubing, and the ability to make a clamp.
The only snag is, who do you trust to intubate you??
Innovation a bed for treating and quarantining patients with
COVID19
A bed was created for a stone infected with Crohn virus, consisting of two rooms. The first room is completely isolated from the second room. The first one resides in the first room. The upper part of the body is the head and chest .
The compressed air into the first chamber through the first air purification and disinfection device, and allows the air inside to be mixed with fresh medical substances that help kill viruses inside the lung.The air is expelled from the first chamber through the scrubbing device. The air is outside. The air is purified by medical solutions or by boiling water.
https://youtu.be/-jbfishz6Nw
https://youtu.be/VgdNT1Zg1s8
https://youtu.be/JGwQhOU7FtM
There is a major flaw in the design as seen in the second photo down in this article. There is not an exhailation valve in the circuit. Without this valve the lungs will not inflate. The gases (air-oxygen mix) will simply bipass the lungs and the endotracheal tube at the “Y” in the circuit. In addition, the “PEEP” valve (positive end expitatory pressure valve) in the circuit is useless in this arrangement.
I can donate my time to and my Prusa printer material if there are parts I could print for anyone making these devices.
I have a 5 ft. X 10 ft. commercial(not home-built) CNC machine sitting idle that I’m sure could make the frame parts (they look like they are made of Plexiglas) within very close tolerances. Would that help? It’s located in my home shop and could be run 24/7.
Is this intubated?
If not, you may need to build a glass shield to prevent aerolizing of the virus, which will infect more patients.
MIT can build a $100 ventilator, but scitechdaily.com can’t properly format a webpage.
Just yesterday, I read an article that a electronically controlled unit has been designed and available to be built quickly for something like $50. It uses solid state printers as well, as well as commercially made valves. Several large hospitals, doctors and ham operators came up with it.
This is totally useless in a clinical setting. The MIT scientists failed on this one.
Reprogram CPAP machines to function as a basic Bi-PAPs and re-engineer the blowers to handle higher ventilator pressures. This does not have the redundancy typically required for intubated ventilation, however it’s better than telling Grandma “good luck surviving the hospital hallway without being able to breathe”. Only reason not happening is because of financial liabilities, companies won’t touch this with a ten-foot pole.
I came here to verify what I already knew.
God Bless MIT for jumping on this. But….
Does anyone here experienced in mass casualty events actually believe that this will work? I’m not being negative, just realistic. This is folly. Let’s just say there are 1k of these vents that worked flawlessly, which they wouldn’t,as Ambu bags aren’t designed for this type of industrial use. Are there a 1k ET tubes, blades, suction units,sedation, monitors, or people that can set them up and run them?
I felt obligated to say something, in the hopes that someone may read this and focus on finding a different solution. I can’t come up with one, and I’ve been to a couple big disasters. A poor analogy would be to hand someone a dry paint brush and say “Here, paint the house!”… “Great, where’s the paint?…”Paint?”
D Douglas – need link to article . . .
This is a good start. How about some sort of printed bellows, I work in GUatemala. Hospitals here do NOT have an abundance of Ambu bags. We would also require some sort of adjustable expiratory valve as to maintain 5 cm pressure so as not to collapse the lungs.
Come on. Such negativity. Have you ever stood in a hallway full of patients with NOTHING! This is going to work. Excellent MIT!
Instead of using an electric motor, why not utilize the hospital air supply. Less heat generated by eliminating the motor. Other electrical components could be eliminated as well using trip switches to control back and forth movement of an air cylinder or maybe an air motor using a gear box reduction to provide constant motion.
You, know with all the model kits I have for my son he never assembled, I could build something better. For example, they say the bag can be hand pumped if needed. I see he has three robot arms, one of which is hydraulic and could easily supply the necessary force. Connect that up to an arduino and a buzzer, and then there is an alarm if it stops operating. Use his air sensor from his wind tunnel, it can check if there is actually proper air flow into and out of the lungs.
But no hospital is going to allow the ventilator to be used… And you cannot hook one up to yourself…
No physician with a patient needing a vent, and no vent available, should pay any attention to a hospital administrator who tried to tell him/her she could not use this device. Lives were saved with the old fixed volume ventilator before sensitive valves were available.
If you’d like to get involved in a local project using the MIT design, can someone please comment what’s required by the FDA to produce a machine that could be used under emergency conditions in a hospital environment? Thank you.
Jan R.
It isn’t negativity. This is reality.People are presenting indisputable facts. You’re a hindrance. Not time for a pep rally, Jan. Let men of science work. I only came back to this podunk website to copy my last post to use for large circulation outlets. Anyone with a emergency medical background would be negligent to *not* present them, and any good engineer will be glad to have them. That’s how engineering works. Any competent anesthesia doc / CRNA / field medic / RT, s#!t, a good HVAC tech, should shoot themselves in the face if they couldn’t slam a simple vent together *if given the proper parts*.
We have a shop in Northeast PA ready to help and we also have a design that we are building on our own to test. We stand ready to help in any way.
Not the first
https://www.ladbible.com/news/news-engineers-use-snorkelling-masks-to-make-ventilators-amid-coronavirus-20200323?c=1584997519697%3Fsource%3Dfacebook&fbclid=IwAR149jdUx888KJehs0LZMjSnE1CIJDCYXBV1wLYCbE4PUY-m0Kgo_bgex50
I did a first pass of the BOM and it’s more than $100.
POST THE PLANS OR POST NOTHING
“PUT UP OR SHUT UP”
Having spent two weeks on a ventilator, as a controls engineer, the biggest problem is constantly control of the air pressures. Pressure must be detected on and through out the cycle of every breath.
It is very painful, and is exhausting to have too little or too much air pressure on the inside of your lungs at any moment.
Especially where your lungs are painfully raw with infection.
We are talking about minute pressure variations that have a complex function through out the breath. A pressure differential at the beginning of a cycle, feels very different mid-cycle and then when your lungs are approaching full.
Also just as in normal breathing, a periodically, you need one deeper or a one more shallow breath.
You often reverse breath direction early.
(This is Not a problem to be solved by linear thinkers who are accel at reproducing canned formula and procedures.)
To have forced air, in/out of your lungs, when your body isn’t calling for it, is very uncomfortable, tiring and doesn’t feel healthy.
Because air is directly compressible it is the force that is important, not the volume.
Pay attention to your breathing, every breath is different. Shallow breaths and various depths of breath are in a complex, non-repeating pattern.
Two requirements:
1) must have very sensitive pressure detection. That is … what are the lung muscles calling for at every moment? (Would be useful to monitor the muscle firing signals)
AND
2) And the most precision control of air flow volume and pressure possible.
Suggest controlling the volume of an air container, by squeezing is way to crude (and painfully cruel).
Two other ways of precisely controlling air pressure come to mind.
1) meter an air supply into a very low resistance diaphragm that is very small percentage of the total breath volume needed, tiny pressure changes are fed through a one-way check valve. A different diaphragm for in and for out breaths (Theses can be double acting)
Some moments the breath is fed as higher pressure burst. Other moments the breath’s pressure differentials are “leaked” slowly.
2) The other approach is to use flexible, small volume, fan blades (flexible so to soften the pressure changes), and are driven by a reversble force controled motor. This let’s you control the occasionial tiny hick-up breaths.
The research begins by putting a mask on that monitors the volume and pressure profile of breathing, and how it changes moment to moment.
Observations recorded of life events, such as: “that take your breath away”, burp, yawn or “give you excitement”.
If you have questions, will attempt to answer.
Caringly,
[email protected]