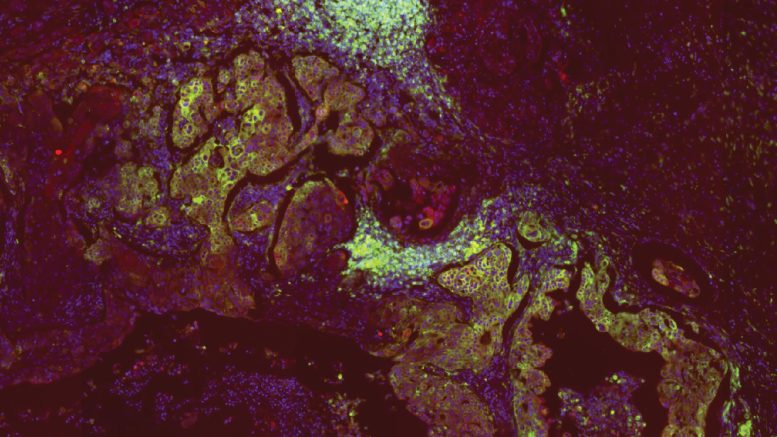
A study at Cold Spring Harbor Laboratory found that breast cancer cells use MHC-II to infiltrate lymph nodes, where they evade immune detection and spread more easily. Disabling MHC-II in cancer cells reversed this effect in mice, slowing metastasis and improving survival.
Lymph nodes act as one of the body’s first lines of defense against disease. From these organic control centers, immune cells are sent into battle against foreign invaders. But somehow, lymph nodes are also the first stop for most metastatic cancers.
“It’s paradoxical,” Cold Spring Harbor Laboratory (CSHL) Assistant Professor Semir Beyaz says. “The cancer goes right in, but the immune cells aren’t doing anything. It’s important to understand what’s going on because this is how cancer takes the whole body hostage.”
Beyaz joined with collaborators from Massachusetts General Hospital to investigate. They found that breast cancer cells trick the immune system with help from a molecule called MHC-II. Future therapeutics targeting this molecule may help slow the cancer’s spread and improve patient outcomes.
How Cancer Exploits Lymph Nodes for Metastasis
“MHC-II acts like breast cancer’s passport,” Beyaz says. “It convinces the lymph node to let the cancer in and protect it. From there, it’s mayhem.”
In other places, like the intestine, MHC-II helps destroy abnormal cells before they become a problem. But breast cancer’s version of MHC-II doesn’t carry the red flags immune cells recognize. So, the lymph node treats it like a false alarm. Beyaz explains:
“Cancer hijacks the lymph node—the police station. The detectives just say, ‘Welcome. Here’s a comfy couch. Here’s a coffee.’ Cancer bribes the neighboring cells. Then it grows. This is what MHC-II is doing in lymph node metastasis.”
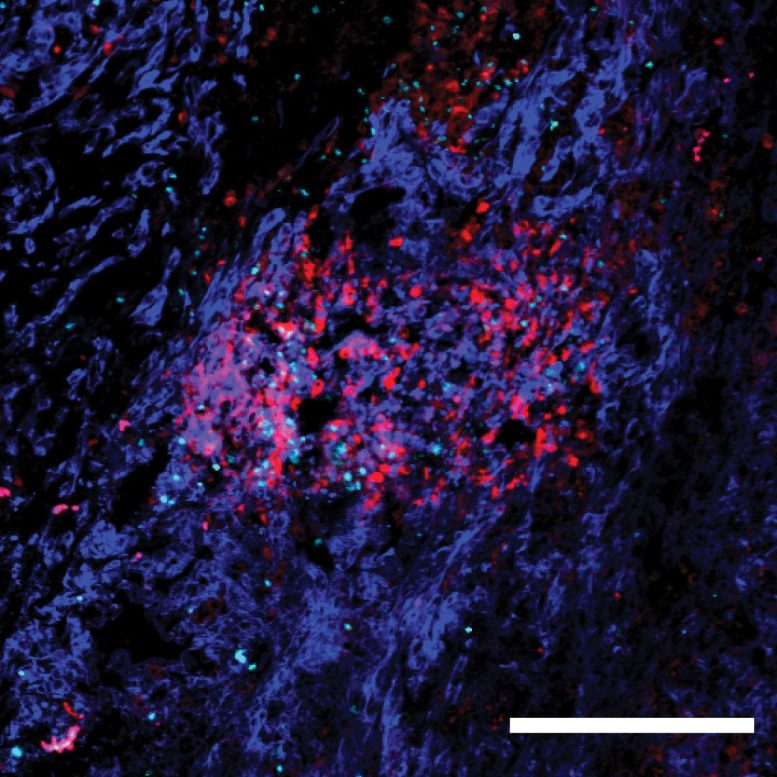
Blocking MHC-II to Slow Cancer’s Spread
The team found that, in mice, higher levels of MHC-II on a subset of cancer cells led to greater immune suppression in lymph nodes. This caused worse metastasis and shorter survival. When they switched off MHC-II production in cancer cells, lymph nodes awoke to the threat. As a result, the cancer couldn’t spread as fast, and the mice lived longer.
“If you get rid of MHC-II in cancer cells, you curb the invasion,” Beyaz explains. “The lymph nodes stop suppressing immune response and reduce cancer’s colonizing abilities.”
Beyaz now hopes to reveal exactly how cancer adapts and spreads. Understanding these mechanisms could bring us closer to new metastasis-blocking therapeutics. But, he cautions, the effectiveness of any potential drug will depend on where cancer first develops.
“For example, in the gut, we see the opposite of what’s happening in breast cancer,” Beyaz explains. “There are context-specific rules, and this tells us there is no one cure-all.”
Over 300,000 people in the U.S. will be diagnosed with breast cancer this year alone. While a long journey lies ahead, Beyaz thinks this research may someday have clinical implications that lead to better therapies and improve patients’ lives.
Reference: “Cancer cell plasticity and MHC-II–mediated immune tolerance promote breast cancer metastasis to lymph nodes” by Pin-Ji Lei, Ethel R. Pereira, Patrik Andersson, Zohreh Amoozgar, Jan Willem Van Wijnbergen, Meghan J. O’Melia, Hengbo Zhou, Sampurna Chatterjee, William W. Ho, Jessica M. Posada, Ashwin S. Kumar, Satoru Morita, Lutz Menzel, Charlie Chung, Ilgin Ergin, Dennis Jones, Peigen Huang, Semir Beyaz and Timothy P. Padera, 21 June 2023, Journal of Experimental Medicine.
DOI: 10.1084/jem.20221847
The study was funded by the National Institutes of Health, a Rullo Family MGH Research Scholar Award, the Oliver S. and Jennie R. Donaldson Charitable Trust, the G. Harold and Leila Y. Mathers Charitable Foundation, the Mark Foundation For Cancer Research, the Chan Zuckerberg Initiative, the Silicon Valley Community Foundation, the STARR Cancer Consortium, Cold Spring Harbor Laboratory, Northwell Health, New York Genome Center, Deutsche Forschungsgemeinschaft Walter Benjamin Programme, and the Agency for Science, Technology and Research (A*STAR).
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.