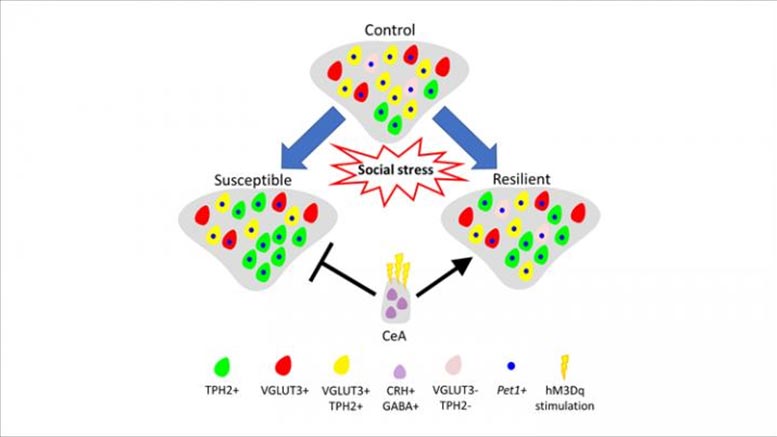
Model of neurotransmitter plasticity in the ventral dorsal raphe in response to chronic social stress. Credit: Prakash et al., JNeurosci 2019
Changes in neurotransmission may explain intrinsic immunity to depression.
Rats susceptible to anhedonia, a core symptom of depression, possess more serotonin neurons after being exposed to chronic stress, but the effect can be reversed through amygdala activation, according to new research in JNeurosci.
Some people are resistant to depression and anhedonia, or lack of pleasure, even when exposed to chronic stress. To measure susceptibility to anhedonia, Prakash et al. trained rats to activate an electrode that stimulated reward circuits in their brain, causing feelings of pleasure. The rats experienced social stress once a day and then were given access to self-stimulation fifteen minutes later. In rats susceptible to anhedonia, the stress dramatically increased the intensity of stimulation needed to feel pleasure, while it had little effect on the resilient rats.
Compared to the resilient rats, the susceptible rats had more serotonin neurons in the ventral part of their dorsal raphe nucleus, an area of the brain involved in regulating stress and reward. This increase is due to the recruitment of non-serotonin signaling neurons. When the researchers activated neurons in the central amygdala to prevent the increase in serotonin signaling, the rats experienced reduced effects from social stress.
Understanding the molecular thumbprint of depression susceptibility could lead to treatments that induce resilience in the face of chronic stress.
Reference: “Serotonergic Plasticity in the Dorsal Raphe Nucleus Characterizes Susceptibility and Resilience to Anhedonia” by Nandkishore Prakash, Christiana J. Stark, Maria N. Keisler, Lily Luo, Andre Der-Avakian and Davide Dulcis, 15 January 2020, JNeurosci.
DOI: 10.1523/JNEUROSCI.1802-19.2019







Be the first to comment on "Stress Doesn’t Always Cause Depression – Here’s Why"